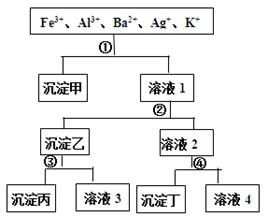
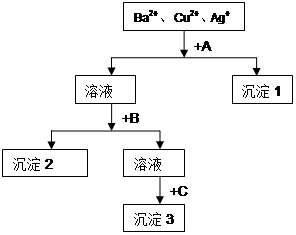
��Ŀ����
ij��Һ�н����±������е�5�����ӣ�������ˮ�ĵ��뼰���ӵ�ˮ�⣩�������ӵ����ʵ�����Ϊ1mol��
������ԭ��Һ�м���KSCN��Һ�������Ա仯��������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䡣��������Һ�м���BaCl2��Һ���а�ɫ���������Իش��������⡣
��1��������ԭ��Һ�м�����������ᣬ�ټ���KSCN��Һ�������� ��
��2��ԭ��Һ�к��е��������� ��
��3����ԭ��Һ�м������������ᣬ������Ӧ�����ӷ���ʽΪ ��
��4����ԭ��Һ�м���������NaOH��Һ����ַ�Ӧ���ˡ�ϴ�ӡ����գ��������ù�����������ƽ��������Ϊ ��
| ������ | SO42-��NO3-��Cl- |
| ������ | Fe3+��Fe2+��NH4+��Cu2+��Al3+ |
������ԭ��Һ�м���KSCN��Һ�������Ա仯��������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䡣��������Һ�м���BaCl2��Һ���а�ɫ���������Իش��������⡣
��1��������ԭ��Һ�м�����������ᣬ�ټ���KSCN��Һ�������� ��
��2��ԭ��Һ�к��е��������� ��
��3����ԭ��Һ�м������������ᣬ������Ӧ�����ӷ���ʽΪ ��
��4����ԭ��Һ�м���������NaOH��Һ����ַ�Ӧ���ˡ�ϴ�ӡ����գ��������ù�����������ƽ��������Ϊ ��
��1����Һ��ΪѪ��ɫ ��2��Fe2+��Cu2+
��3��3Fe2++4H++NO3-��3Fe3++NO��+2H2O ��4��160.0g
��3��3Fe2++4H++NO3-��3Fe3++NO��+2H2O ��4��160.0g
�������������ԭ��Һ�м���KSCN��Һ�������Ա仯����˵����Һ��û�������ӡ�����ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䣬�����Һ��һ�����������ӡ��ܲ������ݵ�ֻ����NO3�������������±���ԭ����NO�����һ�������л�ԭ�������������ӣ�ͬʱ������NO3������������Һ�м���BaCl2��Һ���а�ɫ�������ɣ���˵��������SO42-���������ӵ����ʵ�����Ϊ1mol���������Һ�ĵ����Կ�֪����Һ��һ�������������ӡ�����ֻ�ܺ���5�����ӣ�����������ӵĵ������4mol��֪������һ����������ͭ���ӡ�
��1��������������NO3���ܰ�Fe2����������Fe3��������������ԭ��Һ�м�����������ᣬ�ټ���KSCN��Һ����������Һ��ΪѪ��ɫ��
��2���������Ϸ�����֪��ԭ��Һ�к��е���������Fe2+��Cu2+��
��3���������ᣬ���������Ե�NO3-�ͻ�ԭ�Ե�Fe2+����������ԭ��Ӧ����Ӧ�����ӷ���ʽΪ3Fe2++4H++NO3-��3Fe3++NO��+2H2O��
��4��ԭ��Һ��������������Fe2+��Cu2+������ԭ��Һ�м���������NaOH��Һ����ַ�Ӧ����ˣ�ϴ�ӣ����������أ��õ��Ĺ�����CuO��Fe2O3��������������ӵ����ʵ�����Ϊ1mol��֪��m��CuO����1mol��80g/mol=80g��m��Fe2O3����0.5mol��160g/mol��80g�����ù��������Ϊ80g+80g��160.0g��

��ϰ��ϵ�д�
 ���㼤�������100�ִ��Ծ�ϵ�д�
���㼤�������100�ִ��Ծ�ϵ�д� ��Ȥ������ҵ���ϿƼ�������ϵ�д�
��Ȥ������ҵ���ϿƼ�������ϵ�д�
�����Ŀ

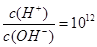 ����Һ�У�
����Һ�У� 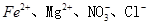
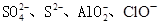
 NH3�� + H2O��NH3��ʹʪ��ĺ�ɫʯ����ֽ��������ij��Һ�п��ܺ�������6�������е�ij���֣�Na+��NH4+��K+��Cl����SO42����CO32����Ϊȷ����Һ��ɽ�������ʵ�飺��1��ȡ200 mL������Һ����������BaCl2��Һ����Ӧ�������ˡ�ϴ�ӡ�����ó���4.30g��������м�����������ᣬ��2.33g�������ܡ���2����1������Һ�м���������NaOH��Һ�����ȣ�������ʹʪ���ɫʯ����ֽ����������1.12L���ѻ���ɱ�״�����ٶ�����������ȫ���ݳ������ɴ˿��Եó�����ԭ��Һ��ɵ���ȷ������
NH3�� + H2O��NH3��ʹʪ��ĺ�ɫʯ����ֽ��������ij��Һ�п��ܺ�������6�������е�ij���֣�Na+��NH4+��K+��Cl����SO42����CO32����Ϊȷ����Һ��ɽ�������ʵ�飺��1��ȡ200 mL������Һ����������BaCl2��Һ����Ӧ�������ˡ�ϴ�ӡ�����ó���4.30g��������м�����������ᣬ��2.33g�������ܡ���2����1������Һ�м���������NaOH��Һ�����ȣ�������ʹʪ���ɫʯ����ֽ����������1.12L���ѻ���ɱ�״�����ٶ�����������ȫ���ݳ������ɴ˿��Եó�����ԭ��Һ��ɵ���ȷ������
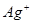 ��
��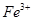 ��
��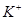 ��
��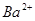 ��
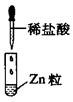
��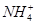 �������ӡ�ijͬѧ��������ʵ�飺
�������ӡ�ijͬѧ��������ʵ�飺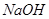 ��Һ����Һ�ʼ��ԣ����ȣ��ɲ���ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����塣
��Һ����Һ�ʼ��ԣ����ȣ��ɲ���ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����塣