��Ŀ����
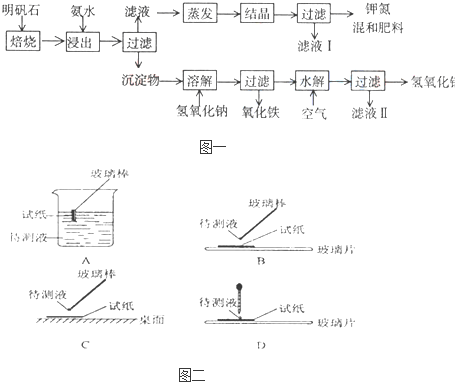
����ʯ����ȡ�طʺ�������������Ҫԭ�ϣ�����ʯ����ɺ��������ƣ�����������������������������ʡ�����ʵ�鲽������ͼ��ʾ��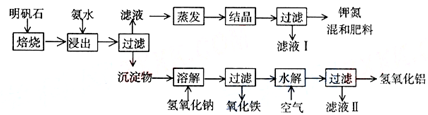
��������ͼʾ�����������գ�
��1������ʯ���պ���ϡ��ˮ����������500mLϡ��ˮ��ÿ������39.20g������ҪȡŨ��ˮ��ÿ������251.28g����__________mL���ù��Ϊ_______mL��Ͳ��ȡ��
��2����ˮ������õ���������ϵ�����ˣ���Һ�г�K����SO42���⣬���д�����NH4��������NH4���ķ�����______________________________________________��
��3��д�����������������ʵĻ�ѧʽ________________________________________��
��4����ҺI�ijɷ���ˮ��______________��
��5��Ϊ�ⶨ��Ϸ���K2SO4��(NH4)2SO4�мصĺ��������������в��裺
�ٳ�ȡ�ص�������������ˮ����������______��Һ��������ɫ������
��___________��__________��_________(������дʵ���������)��
����ȴ�����ء�
��6��������Ϊmg�����������ʵ���Ϊnmol����������K2SO4�����ʵ���Ϊ��___________mol���ú���m��n�Ĵ���ʽ��ʾ����
��1��78 ��100
��2��ȡ��Һ����������NaOH�����ȣ����ɵ�������ʹ��ʪ�ĺ�ɫʯ����ֽ���������������֣�
��3��Al(OH)3��Al2O3��Fe2O3
��4��K2SO4��(NH4)2SO4
��5��BaCl2�����ˣ�ϴ�ӣ�����
��6��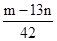
����