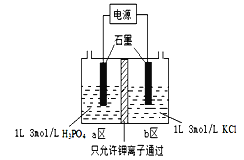
��Ŀ����
����Ŀ���Ķ�����֪ʶ���ش�1������2�����⡣
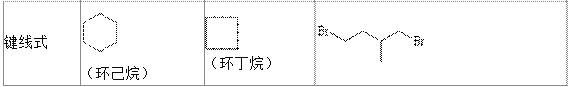
��.�ṹ��û�л�״�������������������������ʽ��CnH2n+2��nΪ����������������ÿ����2��̼�������Ȼͬʱ������1��̼̼�������������ؼ���˫������������Ҳ���������ӻ�״��������Ϊ������һ�������Ͷȣ���ϣ����ĸ����ʾ���ֳ���ȱ��ָ�����������ϩ����=1��������=4��
��.���л�������У���ij��̼ԭ����������4����ͬ��ԭ�ӻ�ԭ���ţ���̼ԭ�ӽв��Գ�̼ԭ�ӣ�ͨ�����Ǻű����*C��������������Ĺ�ѧ���ԡ�
��1���ݱ�������������״��˹��ϳ���һ���п������ԵĻ�����Depudecin���������������������������ṹ��ͼ��ʾ��

����д�����ֻ�����ķ���ʽ________________���û��������Ϊ____________��
��������ӵĽṹ����__________�����Գ�̼ԭ�ӣ�������Ŀ�и����Ľṹ��ʾ����*�����DZ�ʾ������
��2���½��ϳɵ�ij�л��ᆳ�ⶨ���й�ѧ���ԣ���ṹʽ��ͼ��ʾ��

��Ҫʹ���л��ﲻ�پ��й�ѧ���ԣ���ɲ�����Щ��ѧ��������ֱ��г���__________________��__________________��__________________��д�����֣���
���𰸡�C11H16O446 �ӳ���ȥ��ˮ�⡣
�ӳ���ȥ��ˮ�⡣
��������
��1���ٸ����л���̼ԭ�ӳɼ��ص㣬���л���ķ���ʽΪC11H16O4��������Hԭ��Ϊһ�������Ͷȣ��ó���һ����Ϊһ�������Ͷȣ�һ��˫��Ϊһ�������Ͷȣ����ݽṹ��ʽ�����л���IJ����Ͷ�Ϊ4���ڸ���������Ϣ������̼ԭ��Ϊ���Գ�̼ԭ�ӣ�����̼ԭ������4����ͬ��ԭ�ӻ�ԭ���ţ������ ����6�����Գ�̼ԭ�ӣ���2����������̼ԭ�ӵĶ��壬���У�CH2OH�ͣ�CHO�ġ�CH���е�CΪ����̼ԭ�ӣ����������ѧ���ԣ���Ҫ������H2�ļӳɷ�Ӧ������ˮ�ⷴӦ��������Ӧ����ȥ��Ӧ��
����6�����Գ�̼ԭ�ӣ���2����������̼ԭ�ӵĶ��壬���У�CH2OH�ͣ�CHO�ġ�CH���е�CΪ����̼ԭ�ӣ����������ѧ���ԣ���Ҫ������H2�ļӳɷ�Ӧ������ˮ�ⷴӦ��������Ӧ����ȥ��Ӧ��

����Ŀ��(1)2017���п�Ժij�о��Ŷ�ͨ�����һ������Na-Fe3O4/HZSM-5��ܸ��ϴ������ɹ�ʵ����CO2ֱ�Ӽ�����ȡ����ֵ���ͣ����о��ɹ�������Ϊ��CO2��ת�������ͻ���Խ�չ����
��֪��H2(g)+1/2O2(g)=H2O(l) ��H1 = ��aKJ/mol
C8H18(1)+25/2O2(g)=8CO2(g)+9H2O(1) ��H2= ��bKJ/mol
��д��25����101kPa�����£�CO2��H2��Ӧ��������(��C8H18��ʾ)���Ȼ�ѧ����ʽ_________________________________��
(2)����CO2��H2Ϊԭ�ϣ��ں��ʵĴ���(��Cu/ZnO����)�����£�Ҳ�ɺϳ�CH3OH���漰�ķ�Ӧ�У�
�ף�CO2(g)+3H2(g) ![]() CH3OH(g)+H2O(g) ��H= �� 53.7kJ��mol-1 ƽ�ⳣ��K1
CH3OH(g)+H2O(g) ��H= �� 53.7kJ��mol-1 ƽ�ⳣ��K1
�ң�CO2(g)+H2(g) ![]() CO(g)+H2O(g) ��H= + 41.2kJ��mol-1 ƽ�ⳣ��K2
CO(g)+H2O(g) ��H= + 41.2kJ��mol-1 ƽ�ⳣ��K2
��CO(g)+2H2(g) ![]() CH3OH(g)��ƽ�ⳣ��K=______(�ú�K1��K2�ı���ʽ��ʾ)���÷�Ӧ��H_____0(��������������С����)��
CH3OH(g)��ƽ�ⳣ��K=______(�ú�K1��K2�ı���ʽ��ʾ)���÷�Ӧ��H_____0(��������������С����)��
�����CO2ת��ΪCH3OHƽ��ת���ʵĴ�ʩ��___________(��д����)��
�۴����ͷ�Ӧ��ϵ�Ĺ�ϵ��������Կ�Ĺ�ϵһ�������и߶ȵ�ѡ���ԡ���������ʵ�飬����CO2��H2��ʼͶ�ϱȾ�Ϊ1��2.2��������ͬ��Ӧʱ��(t1min)��
�¶�(K) | ���� | CO2ת����(%) | �״�ѡ����(%) | �ۺ�ѡ�� |
543 | Cu/ZnO���װ����� | 12.3 | 42.3 | A |
543 | Cu/ZnO����Ƭ���� | 11.9 | 72.7 | B |
553 | Cu/ZnO���װ����� | 15.3 | 39.1 | C |
553 | Cu/ZnO����Ƭ���� | 12.0 | 70.6 | D |
�ɱ����е����ݿ�֪����ͬ�¶��²�ͬ�Ĵ�����CO2��ת��ΪCH3OH��ѡ����������Ӱ�죬�����ϱ��������ݽ�Ϸ�Ӧԭ������������ѡ��Ϊ___________(����ĸ����)��
(3)��CO��H2Ϊԭ�Ϻϳ��״��ķ�ӦΪ��CO(g)+2H2(g)![]() CH3OH(g)���������Ϊ2L�����������ܱ����������������У��ֱ���1molCO��2molH2�����������ķ�Ӧ�¶ȷֱ�ΪT1��T2��T3�Һ㶨���䡣��ͼΪ���������еķ�Ӧ�����е�5minʱH2���������ʾ��ͼ��������һ��������Ӧһ���ﵽƽ��״̬��
CH3OH(g)���������Ϊ2L�����������ܱ����������������У��ֱ���1molCO��2molH2�����������ķ�Ӧ�¶ȷֱ�ΪT1��T2��T3�Һ㶨���䡣��ͼΪ���������еķ�Ӧ�����е�5minʱH2���������ʾ��ͼ��������һ��������Ӧһ���ﵽƽ��״̬��
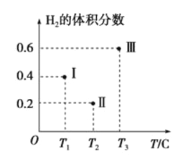
��0��5minʱ��������������CH3OH��ʾ�Ļ�ѧ��Ӧ����Ϊ_________________��
������������һ���ﵽƽ��״̬��������________(��д��������)��