��Ŀ����
I��������Ӳ�ȴ���ʴ������Ҫ�ĺϽ���ϡ�
��1����̬��ԭ�ӵļ۵��ӹ����ʾʽΪ________________��CrO2Cl2������Ϊ���ɫҺ�壬����CCl4���ܣ��ݴ��ж�CrO2Cl2�ľ�����������_________________��
��2�������CrCl3��6H2O����λ��Ϊ6���������ֲ�ͬ��ɡ���ͬ��ɫ�Ĺ��壬����һ��Ϊdz��ɫ���壬�ù�����������������Һ��Ӧʱ��0.5mol���������1mol�Ȼ���������������dz��ɫ�������������ȣ�Cl-��H2O��Ϊ_____________��
II.�黯��Ϊ�������뵼����ϣ������ṹ��ͼ��ʾ��
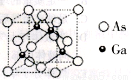
��3���黯�ؿ���(CH3)3Ga��AsH3��700���·�Ӧ�Ƶã���Ӧ�Ļ�ѧ����ʽΪ__________��
��4��AsH3���ӵ����幹��Ϊ__________������Asԭ�ӵ��ӻ���ʽ��______��1��(CH3)3Ga�����к���_________���Ҽ�����ĵ�һ������_______���>����<�����صĵ�һ�����ܡ�
��5���黯�ؾ���������ص����ԭ�Ӻ˼��Ϊa nm���黯�ص�Ħ������Ϊbg��mol-1�������ӵ�����ֵΪNA�����黯�ؾ�����ܶȱ���ʽ��_____________g��cm-3��
 �ǻ�С��ϰϵ�д�
�ǻ�С��ϰϵ�д�����ʵ�鷽���У����ܴﵽʵ ��Ŀ�ĵ���
��Ŀ�ĵ���
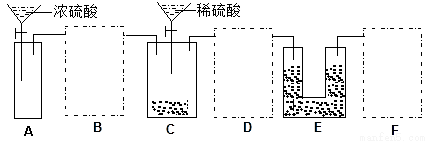
ѡ�� | ʵ��Ŀ�� | ʵ�鷽�� |
A | ������������NO2 | �ֱ�ͨ����������Һ�У���������ɫ�������������� |
B | ֤�������ԣ�H2O2��Fe3��ǿ | �������ữ��H2O2��Һ����Fe(NO3)2��Һ�У���Һ���ɫ |
C | ֤������ˮ�������ȷ�Ӧ | �ڴ�������Һ�е����̪��Һ�����Ⱥ��ɫ���� |
D | ֤����������ڳ����ܽ�ƽ�� | ����ɫ����������PbI2��ˮ��������ȡ�ϲ���Һ����NaI���������ɫ���� |
A. A B. B C. C D. D