��Ŀ����
����һƿ��NaCl���ʵ�Na2CO3��Ʒ��ijͬѧ�����һ�ֲⶨ�䴿�ȵķ�����װ������ͼ������A������ʢ����Ʒ10.0 g����Һ©����ʢ��ϡ���ᣬB������Ϊ��ʯ�ҹ��塣
��1����ͬѧ�ظ����������Σ�ÿ�β�������ȷ����װ�ò�©�������ǽ�����ݳ����˽ϴ�ƫ������ڷ�Ӧ����ȫ��װ���ڿ�����Ӱ����Բ��ƣ�������������ܵ�ԭ��______��
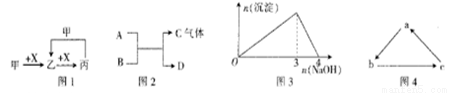
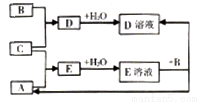
��2��ͨ����ϸ��������ͬѧ���������һ��ʵ��װ������ͼ����ش��й����⣺
���ɹ�ѡ����Լ�����ZnƬ��Ũ���ᡢNaOH��Һ��CaCO3���塢����ˮ��
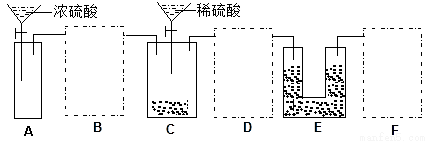
�ٸ���װ��δ����������������Ϊ��Ҫ����װ�õ�����ڻ������ʵļ�ͼ����ע���Լ���______
��Aװ�õĹ��ƿ��Ӧ������Լ���__________����װ�õ�������________________��
�����ʵ�����Eװ����������3.52 g�����Na2CO3�����Ĵ���Ϊ__________��
��3���ⶨ��Na2CO3�������ȵķ����ж��֣��������ò�ͬ�ķ�Ӧԭ�������һ������
Ҫ��д��ʵ��������̺�������Na2CO3���ȵı���ʽ__________�����Լ���ѡ��
�������̰�����ͼʾ������

�ٲ�������Ϊ��

��ʵ��ʱ������Ʒ������Ϊa�ˣ��������ʵ�����Ϊb�ˣ���������Na2CO3���ȵı���ʽΪ��__________��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���HNO3��HF�Ļ��ᴦ��ij������������ϴ��Һ�к���Fe3+��Ni2+��NO3-��F-��Cr2O72-�ȡ���ͼ���ۺ����ø���ϴ��Һ�Ĺ������̣�
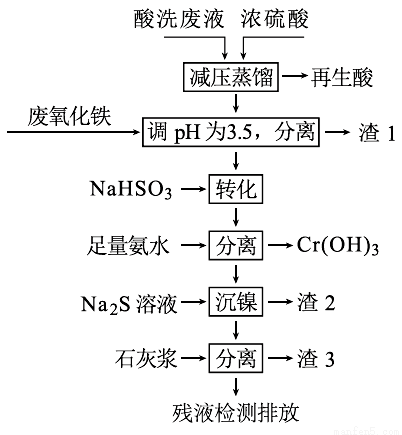
��֪��
�������ӿ�ʼ�����ͳ�����ȫʱ��pH��
Fe3+ | Ni2+ | Cr3+ | |
��ʼ���� | 1.5 | 6.7 | 4.0 |
������ȫ | 3.4 | 9.5 | 6.9 |
��Ni2+��������ˮ�ķ�ӦΪ��Ni2+��6NH3 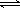 [Ni(NH3)6]2+
[Ni(NH3)6]2+
��1���������к���HNO3,��ȡ��ѹ�����Ŀ��Ϊ____________��
��2������������Ҫ�ɷ�Ϊ��____________��
��3����д����ת����ʱNaHSO3��Cr2O72-������Ӧ���������뻹ԭ�������ʵ���֮�ȣ�________��
��4����֪[Ni(NH3)6]2+Ϊ�ѵ����������ӣ������������ӷ���ʽΪ��____________��
��5������3����Ҫ�ɷ�ΪCaSO4��Ca(OH)2��_____��
��6������⣬���IJ�Һ��c(Ca2+)��0.001 mol•L-1�����Һ��F��Ũ��Ϊ____mg•L-1��______������ϡ������ϡ����ŷű�[��֪Ksp(CaF2)=4��10-11�������ŷű�Ҫ�������Ũ��С��10 mg•L-1]��


 A
A