��Ŀ����
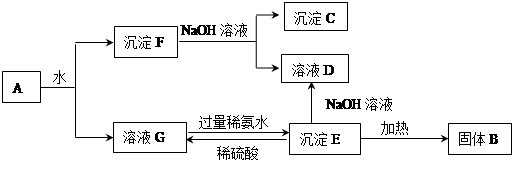
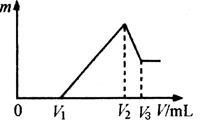
Ϊ�˲ⶨijþ���Ͻ�ijɷ֣�ȡ14.7 g�Ͻ���ȫ����500 mL 3 mol/L�������У��ټ���400 mL 8 mol/L������������Һ��ַ�Ӧ�����ֻ����һ�ֳ���������ڸúϽ�IJⶨ���̵�������ȷ����( )
| A���Ͻ���þ����������Ϊ63.3%��Mg%<100% |
| B���úϽ��к���������������Ϊ5.4 g |
| C���ڲ������������Һ��һ������0.2 mol NaAlO2 |
| D���ڲ������������Һ����1.5 mol Na2SO4 |
AD
���������������ü���ֵ����������ǡ����ȫ��Ӧ����������Ҳǡ����ȫ��Ӧ�����ʱ���ĺ���Ϊ���ֵ��������ת��Ϊƫ�������þ����ת��Ϊ������þ��������ʱ��Һ������ΪNa2SO4��NaAlO2������������غ���n��Na2SO4��=n��H2SO4��="0.5L��" 3 mol/L="1.5" mol�������������غ���n��NaOH��=2n��Na2SO4��+n��NaAlO2��="0.4L" ��8 mol/L=3.2mol���ݴ˼���n��NaAlO2��=0.2mol���ٸ�����ԭ���غ�n��Al��=n��NaAlO2��=0.2mol��m��Al��=5.4g��m��Mg��=9.3g����Mgռ����Ͱٷ���Ϊ63.3%������ѡ��B��C����AD��ȷ��

��ϰ��ϵ�д�
�����Ŀ