��Ŀ����
����Ŀ���������ӷ���ʽ��д��ȷ���ǣ� ��
A.��ʯī���缫���AlCl3��Һ��2Cl-+2H2O![]() 2OH-+H2��+Cl2��
2OH-+H2��+Cl2��
B.Fe(OH)3�������HI��Һ��Fe(OH)3��3H��=Fe3��+3H2O
C.NaNO2��Һ�м�������KMnO4��Һ��2MnO4-+5NO2-+6H��=2Mn2++5NO3-+3H2O
D.NaHSO4��Һ��Ba(OH)2��Һ��ϣ�SO42-+Ba2+=BaSO4��
���𰸡�C
��������
A. ��ʯī���缫���AlCl3��Һ����������Cl-ʧȥ���ӱ�ΪCl2������������Һ�е�H+�õ����ӱ�ΪH2��������OH-����Һ�е�Al3+��Ӧ����Al(OH)3���������ܴ������棬A����
B. Fe(OH)3�������HI��Һ����������ԭ��Ӧ��������Fe3+��I-���һ������������ԭ��Ӧ�����ܴ������棬B����
C. NO2-���л�ԭ�ԣ�����KMnO4��Һ���������ԣ����ᷢ��������ԭ��Ӧ��ʽ�ӷ��Ϸ�Ӧʵ�ʣ���ѭ�����غ㡢����غ㡢ԭ���غ㼰���ʲ��ԭ��C��ȷ��
D. ��Ӧ���γ�BaSO4�����⣬�����������H2O�γɣ�D����
�ʺ���ѡ����C��

 ��������ϵ�д�
��������ϵ�д� ��ӡ�Ļ���ʱ����ϵ�д�
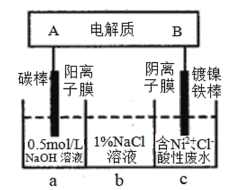
��ӡ�Ļ���ʱ����ϵ�д�����Ŀ����ҵ����ƽ�VOSO4�е�K2SO4��SiO2���ʳ�ȥ�����յõ�V2O5���������£�
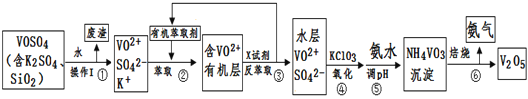
��ش��������⣺
��1�����������÷����ijɷ���____________д��ѧʽ��������I������______��
��2�������������ı仯���̿ɼ�Ϊ����ʽR��ʾVO2+��HA��ʾ�л���ȡ������
R2(SO4)n(ˮ��)+ 2nHA���л��㣩![]() 2RAn���л��㣩 + nH2SO4 (ˮ��)
2RAn���л��㣩 + nH2SO4 (ˮ��)
������ȡʱ��������������ԭ����____________________________��
����X�Լ�Ϊ___________________��
��3���������ӷ���ʽΪ________________________��
��4��25��ʱ��ȡ����������������õ��������ʺ���ҺpH֮���ϵ���±���
pH | 1��3 | 1��4 | 1��5 | 1��6 | 1��7 | 1��8 | 1��9 | 2��0 | 2��1 |
��������% | 88��1 | 94��8 | 96��5 | 98��0 | 98��8 | 98��8 | 96��4 | 93��1 | 89��3 |
����ϱ�����ʵ�������У����м��백ˮ��������Һ�����pHΪ__________��
����������Ϊ93��1%ʱ������Fe(OH)3����������Һ��c(Fe3+)<_____________��
(��֪��25��ʱ��Ksp[Fe(OH)3]=2��6��10-39)
��5���ù��������У�����ѭ�����õ�������______________��_______��
����Ŀ��.�װ�Ǧ��(CH3NH3PbI3)������ȫ��̬���ѿ�����̫���ܵ�ص��������������ʿ��ɼװ�(CH3NH2)��PbI2 �� HI Ϊԭ�����ϳɡ���ش��������⣺
(1)��ȡ�װ��ķ�ӦΪ CH3OH(g)+NH3(g)==CH3NH2(g)+H2O(g)����֪�÷�Ӧ����ػ�ѧ���ļ����������±���ʾ��
���ۼ� | C-O | H-O | N-H | C-N |
����/(kJ | 351 | 463 | 393 | 293 |
��÷�Ӧ����H=_____kJ��mol��1
(2)��ҵ������ˮú���ϳɼ״��ķ�ӦΪ CO(g)+2H2(g) ![]() CH3OH(g) ��H<0��һ���¶��£� �����Ϊ 2L ���ܱ������м��� CO �� H2��5min ĩ��Ӧ�ﵽ��ѧƽ��״̬����ø����Ũ�����±���ʾ��
CH3OH(g) ��H<0��һ���¶��£� �����Ϊ 2L ���ܱ������м��� CO �� H2��5min ĩ��Ӧ�ﵽ��ѧƽ��״̬����ø����Ũ�����±���ʾ��
���� | CO | H2 | CH3OH |
Ũ��/(mol | 0.9 | 1.0 | 0.6 |
��0��5min �ڣ��� CO ��ʾ��ƽ����Ӧ����Ϊ_____��
�ڼ��ܼӿ췴Ӧ���ʣ������������ת���ʵĴ�ʩ��_____(��һ������)��
����˵��������Ӧ�Ѵﻯѧƽ��״̬����_____(����ĸ)��
A��v�� (CO)=2v�� (H2)
B����������ܶȱ��ֲ���
C����Ӧ������ѹǿ���ֲ���
D����������ƽ��Ħ����������
(3)PbI2 ���� Pb3O4 �� HI ��Ӧ�Ʊ�����Ӧ�Ļ�ѧ����ʽΪ_____��
(4)������ PbI2 ������Һ�� c(I-)=2.0��10-3mol��L-1���� Ksp(PbI2)=________����֪Ksp(PbS)=4.0��10-28����Ӧ PbI2(s)+2-(aq) ![]() PbS(s)+2I-(aq)��ƽ�ⳣ��K=_______________________��
PbS(s)+2I-(aq)��ƽ�ⳣ��K=_______________________��
(5)HI �ķֽⷴӦ���ߺ�Һ�෨�Ʊ� HI �ķ�Ӧ���߷ֱ���ͼ 1 ��ͼ 2 ��ʾ��
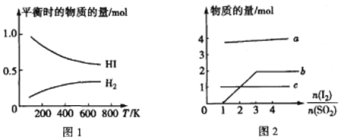
�ٷ�Ӧ H2(g)+I2(g)![]() 2HI(g)����H__________(����>������<")0��
2HI(g)����H__________(����>������<")0��
�ڽ� SO2 ͨ���ˮ�лᷢ����Ӧ��SO2+I2+2H2O==4H++SO42-+2I-��I2+I- ![]() I3-��ͼ 2 ������ b ������������_____(��������)��
I3-��ͼ 2 ������ b ������������_____(��������)��