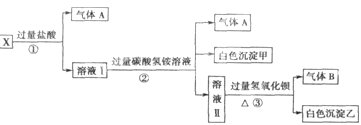
��Ŀ����
ij���ͭ�������ַ�ˮ��Ҫ������һ�ַ�ˮ�к���CN-���ӣ���һ�ַ�ˮ�к���Cr2O72-���ӣ��ó��ⶨ��ͼ��ʾ�ķ�ˮ�������̡�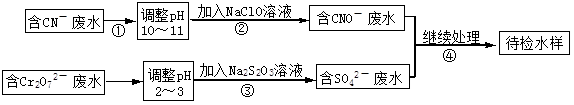 �ش��������⣺
�ش��������⣺
��1������ڷ�����Ӧ�����ӷ���ʽ�ɱ�ʾ���£�aCN��+bClO��+2cOH��=dCNO��+eN2��+fCO32��+bCl��+cH2O���������ӷ���ʽ���ܵ���ƽϵ���ж��飬��ش�
�ٷ���ʽ��e : f��ֵΪ ����ѡ���ţ���
| A��1 | B��1/2 | C��2 | D������ȷ�� |
������Ӧ��ת��0.6mol���ӣ������ɵ������ڱ���µ������ ��
��2��������з�Ӧʱ��ÿ0.4molCr2O72-ת��2.4mol�ĵ��ӣ��÷�Ӧ�����ӷ���ʽΪ ��
��3��ȡ��������ˮ�����Թ��У��ȼ���NaOH��Һ���۲쵽����ɫ�������ɣ���������NaOH��Һ��ֱ�����ٲ�����ɫ����Ϊֹ���ټ���Na2S��Һ���к�ɫ�������ɣ�����ɫ�������٣� �������ӷ���ʽ��ʾ����������ɫ�仯��ԭ��
�ٲ�����ɫ���������ӷ���ʽΪ ���ں��ֱ��ɫ���������ӷ���ʽΪ ��
��4��ͭ���������ϵ�dz����е���ɫ��������֪�����£�����Һ��Cu2+�ȶ���Cu+�������������·�����2Cu+=" Cu+" Cu2+�������+1��ͭ�Ļ�������������磺Cu2O��CuI��CuCl��CuH�ȡ�
��д��CuH�ڹ���ϡ���������������ɵ����ӷ���ʽ ��
�ڽ�CuH�ܽ���������ϡ�����У�������з�Ӧ�Ļ�ѧ����ʽ:
��1����B��6��1.12L
��2��4Cr2O72����3S2O32����26H����6SO42����8Cr3����13H2O
��3����Cu2����2OH����Cu(OH)2��
��Cu(OH)2(s)��S2��(aq)��CuS(s) ��2OH��(aq)
��4����2CuH��2H����Cu��Cu2����2H2��
��6CuH��16HNO3��6Cu(NO3)2��3H2����4NO����8H2O
���������������1�������������غ㣬��֪ѡB�������ݵ���غ��֪Ϊ6
���㣺������ԭ���й�֪ʶ��

��ϰ��ϵ�д�
 100�ִ�����ĩ���ϵ�д�
100�ִ�����ĩ���ϵ�д� ��У���˿��ֿ���ϵ�д�
��У���˿��ֿ���ϵ�д�
�����Ŀ
�ֱ���һ���Լ������������л�����������ʳ�ȥ��������Ϊ��������ʣ�
| ���� | �������Լ� | �й����ӷ���ʽ |
| ��1��HNO3(H2SO4) | | |
| ��2��Cu(Fe) | | |
| ��3��NaCl(Na2CO3) | | |
���������������ļӿ죬�����С��װʳƷ�ѱ��㷺���ܡ�Ϊ���ӳ�ʳƷ�ı����ڣ���ֹʳƷ�ܳ�����֬ʳƷ�������ʣ��ڰ�װ����Ӧ����Ļ�ѧ������
| A����ˮ����ͭ������ | B���轺���������� |
| C��ʳ�Ρ��������� | D����ʯ�ҡ�ʳ�� |
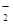 ��MnO
��MnO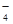 ��CO
��CO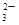 ��SO
��SO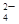 �е�������������ɣ�ȡ��Һ������ͼʵ�飺
�е�������������ɣ�ȡ��Һ������ͼʵ�飺