��Ŀ����
ij��ѧѧϰС���ͬѧ��̽���ⶨ���ᾧ�壨H2C2O4?xH2O����x��ֵ��ͨ���������ϸ�С��ͬѧ��֪������������ˮ����ˮ��Һ����������KMnO4��Һ������Ӧ2MnO4-+5H2C2O4+6H+�T2Mn2++10CO2��+8H2O������ͬѧ���ø÷�Ӧԭ������˵ζ��ķ����ⶨxֵ��
�ٳ�ȡ1.260g�����ᾧ�壬�����Ƴ�100.00mLˮ��ҺΪ����Һ��
��ȡ25.00mL����Һ������ƿ�У��ټ���������ϡH2SO4
����Ũ��Ϊ0.1000mol/L��KMnO4����Һ���еζ���ʵ���¼�й��������£�
��ش�
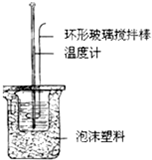
��1���ζ�ʱ����KMnO4��Һװ����ͼ�е�______����ס����ҡ����ζ����У�
��2����ʵ��ζ��ﵽ�յ�ı�־������______��
��3��ͨ���������ݣ������x=______��
��4�������ζ��յ�ʱ���ӵζ��̶ܿȣ����ɴ˲�õ�xֵ��______���ƫ����ƫС�����䡱����ͬ����
�����ζ�ʱ���õ�KMnO4��Һ����ö�����Ũ�ȱ�С�����ɴ˲�õ�xֵ��______��

�ٳ�ȡ1.260g�����ᾧ�壬�����Ƴ�100.00mLˮ��ҺΪ����Һ��
��ȡ25.00mL����Һ������ƿ�У��ټ���������ϡH2SO4
����Ũ��Ϊ0.1000mol/L��KMnO4����Һ���еζ���ʵ���¼�й��������£�
| �ζ����� | ���������Һ�����mL�� | 0.1000mol/LKMnO4����Һ�����mL�� | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| ��һ�� | 25.00 | 0.00 | 10.02 |
| �ڶ��� | 25.00 | 0.22 | 11.32 |
| ������ | 25.00 | 1.56 | 11.54 |
��1���ζ�ʱ����KMnO4��Һװ����ͼ�е�______����ס����ҡ����ζ����У�
��2����ʵ��ζ��ﵽ�յ�ı�־������______��
��3��ͨ���������ݣ������x=______��
��4�������ζ��յ�ʱ���ӵζ��̶ܿȣ����ɴ˲�õ�xֵ��______���ƫ����ƫС�����䡱����ͬ����
�����ζ�ʱ���õ�KMnO4��Һ����ö�����Ũ�ȱ�С�����ɴ˲�õ�xֵ��______��

��1����ΪKMnO4����ǿ�����ԣ��ḯʴ�ܣ���Ӧ����ʽ�ζ���ʢװ���ʴ�Ϊ���ף�
��2����KMnO4��Һ��������ɫ��Ϊָʾ���жϵζ��յ�ʱ���ٵμ�KnO4��Һʱ����Һ������ɫ��Ϊ��ɫ��
�ʴ�Ϊ�����������һ��KMnO4��Һʱ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ���ɫ������ζ��յ㣻
��3����3��2MnO4-+5H2C2O4 +6H+�T2Mn2++10CO2��+8H2O
2 5
0.1000mol/L��0.01L 0.0025mol
25.00mL����Һ�к���0.0025molH2C2O4��100.00mL����Һ�к���0.01molH2C2O4��0.01molH2C2O4������Ϊ0.01mol��90g/mol=0.9g������1.260g�����ᾧ����ˮ�����ʵ���Ϊ1.260g-0.9g=0.36g�������ʵ���Ϊ0.02mol����x=2��
���ζ��յ�ʱ���ӵζ��ܶ�������������������KMnO4��Һ�����ƫС���ɴ�����n��H2C2O4��ƫС����n��H2O��ƫ��xƫ��
ͬ������KMnO4��Һ���ʣ������������ƫ������xֵƫС��
�ʴ�Ϊ��2��ƫ��ƫС��
��2����KMnO4��Һ��������ɫ��Ϊָʾ���жϵζ��յ�ʱ���ٵμ�KnO4��Һʱ����Һ������ɫ��Ϊ��ɫ��
�ʴ�Ϊ�����������һ��KMnO4��Һʱ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ���ɫ������ζ��յ㣻
��3����3��2MnO4-+5H2C2O4 +6H+�T2Mn2++10CO2��+8H2O
2 5
0.1000mol/L��0.01L 0.0025mol
25.00mL����Һ�к���0.0025molH2C2O4��100.00mL����Һ�к���0.01molH2C2O4��0.01molH2C2O4������Ϊ0.01mol��90g/mol=0.9g������1.260g�����ᾧ����ˮ�����ʵ���Ϊ1.260g-0.9g=0.36g�������ʵ���Ϊ0.02mol����x=2��
���ζ��յ�ʱ���ӵζ��ܶ�������������������KMnO4��Һ�����ƫС���ɴ�����n��H2C2O4��ƫС����n��H2O��ƫ��xƫ��
ͬ������KMnO4��Һ���ʣ������������ƫ������xֵƫС��
�ʴ�Ϊ��2��ƫ��ƫС��

��ϰ��ϵ�д�
�����Ŀ