��Ŀ����
15��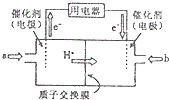 ����������SeO2����һ�����������䱻��ԭ��ĵ��������ܳ�Ϊ������Ⱦ�ͨ����ŨHNO3��H2SO4��Ӧ����SeO2�Ի���Se�����������գ�
����������SeO2����һ�����������䱻��ԭ��ĵ��������ܳ�Ϊ������Ⱦ�ͨ����ŨHNO3��H2SO4��Ӧ����SeO2�Ի���Se�����������գ���1��Se��ŨHNO3��Ӧ�Ļ�ԭ����ΪNO��NO2����NO��NO2�����ʵ���֮��Ϊ1��1��д��Se��ŨHNO3�ķ�Ӧ����ʽSe+2HNO3��Ũ��=H2SeO3+NO��+NO2�����粻�Ը÷�Ӧ��ԭ����ı��������ƣ��÷�Ӧ���ܳ��ֶ�����ƽϵ��������ܡ������ܡ�����˵�����ɸ÷�Ӧ�����ֻ�ԭ������ߵı����ͻ�ԭ�������������Է����ı䣮
��2�����յõ���SeO2�ĺ���������ͨ������ķ����ⶨ��
��SeO2+4KIʮ4HNO3�TSe+2I2+4KNO3ʮ2H2O���� ��I2+2Na2S2O3�TNa2S4O6+2NaI
ʵ���У�ȷ����SeO2��Ʒ0.1600g��������0.2000mol•L-1��Na2S2O3��Һ25.00mL�����ⶨ����Ʒ��SeO2����������Ϊ86.6%��
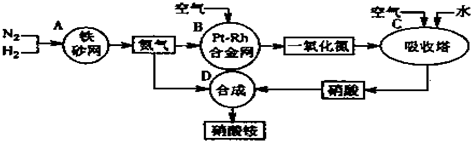
��3��ֱ�Ӽ״�ȼ�ϵ�أ����DMFC��������ṹ������ת���ʸߡ��Ի�������Ⱦ������Ϊ������Դ�����Ʒ��Խ��Խ�ܵ���ע��DMFC�Ĺ���ԭ����ͼ��ʾ��
��ͨ��a����ĵ缫�ǵ�صĸ��������������������缫��ӦΪCH3OH-6e-+H2O=CO2+6H+��
�ڳ����£��ô˵���Զ��Ե缫���0.5L����ʳ��ˮ��������������������������1.12L��������Ϊ��״���µ���������������Һ��pHΪ13��������Һ������仯����
���� ��1������������Ϣ��֪Se��ŨHNO3��Ӧ��Se������Ϊ+4�۵�H2SeO3��HNO3��ԭΪNO��NO2�����õ����غ��������������NO��NO2�����ʵ���֮��Ϊ1��1����������ϵ����Ϊ1��1���ɵ÷���ʽ��������ֻ�ԭ���������
��2�����ݷ�Ӧ�ķ���ʽ��֪��SeO2��2I2��4Na2S2O3������n=cV�������ĵ�n��Na2S2O3�������ݹ�ϵʽ������Ʒ��n��SeO2�����ٸ���m=nM����SeO2������������������Ʒ��SeO2������������
��3���ٸ���ͼ֪������Ĥ�����ӽ���Ĥ����������Һ�����ԣ������������ƶ�����֪��ͨ��a�ĵ缫Ϊ������ͨ��b�ĵ缫Ϊ�����������ϼ״�ʧȥ���ӷ���������Ӧ��
�ڸ��ݵ��صĹ���ԭ���͵缫����ʽ�����㣮
��� �⣺��1������������Ϣ��֪Se��ŨHNO3��Ӧ��Se������Ϊ+4�۵�H2SeO3��HNO3��ԭΪNO��NO2������NO��NO2�����ʵ���֮��Ϊ1��1����������ϵ����Ϊ1��1�������ϵ��Ϊ1�����ݵ���ת���غ��֪��Se��ϵ��Ϊ$\frac{1��3+1��1}{4}$=1���ʷ�Ӧ����ʽΪSe+2HNO3��Ũ��=H2SeO3+NO��+NO2�����粻�Ը÷�Ӧ��ԭ����ı��������ƣ��÷�Ӧ���ܳ��ֶ�����ƽϵ��������÷�Ӧ�����ֻ�ԭ������ߵı����ͻ�ԭ�������������Է����ı䣻
�ʴ�Ϊ��Se+2HNO3��Ũ��=H2SeO3+NO��+NO2�������ܣ��÷�Ӧ�����ֻ�ԭ������ߵı����ͻ�ԭ�������������Է����ı䣻
��2�����ݷ�Ӧ�ķ���ʽ��֪SeO2��2I2��4Na2S2O3�����ĵ�n��Na2S2O3��=0.2000 mol/L��0.025L=0.005mol�����ݹ�ϵʽ������Ʒ��n��SeO2��=0.005mol��$\frac{1}{4}$=0.00125mol����SeO2������Ϊ0.00125mol��111g/mol=0.13875g��������Ʒ��SeO2����������Ϊ$\frac{0.13875g}{0.1600g}$��100%=86.6%���ʴ�Ϊ��86.6%��
��3���ٸ���ͼ֪������Ĥ�����ӽ���Ĥ����������Һ�����ԣ������������ƶ�����֪��ͨ��a�ĵ缫Ϊ������ͨ��b�ĵ缫Ϊ�����������ϼ״�ʧȥ���ӷ���������Ӧ��������ӦʽΪ CH3OH-6e-+H2O=CO2+6H+�������������õ��ӷ�����ԭ��Ӧ���缫��ӦʽΪO2+4e-+4H+=2H2O��
�ʴ�Ϊ������CH3OH-6e-+H2O=CO2+6H+��
���ô˵���Զ��Ե缫��ⱥ��ʳ��ˮ�ĵ缫��ӦΪ��2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$H2��+Cl2��+2OH-�����������ռ�����״���µ�����1.12L��0.05mol��ʱ���������������Ƶ����ʵ�����0.05mol������COH-=$\frac{0.05mol}{0.5L}$=0.1mol/L������PH=13���ʴ�Ϊ��13��
���� ���⿼��������ԭ��Ӧ�������Լ��绯ѧ��֪ʶ�ȣ�Ϊ��Ƶ���㣬�����غ㷨Ϊ���Ĺؼ�������������ԭ��Ӧ��ƽ������Ŀ��飬ע�����ջ���֪ʶ�����գ���Ŀ�ѶȲ���

| A�� | 0.1 mol/L��ˮ�У�c��NH4+��+c��H+��=c��OH-�� | |
| B�� | 0.1 mol/L��NH4Cl��Һ�У�c��NH4+��=c��Cl-�� | |
| C�� | pH=12��NaOH��Һ��pH=12�İ�ˮ�У�c��Na+��=c��NH4+�� | |
| D�� | pH=3�������pH=11�İ�ˮ�������Ϻ�c��OH-��=c��H+�� |
| A�� | 4 | B�� | 4.5 | C�� | 5.5 | D�� | 6 |
| A�� | �٢ڢ� | B�� | �٢ܢ� | C�� | �٢ڢ� | D�� | �ڢܢ� |
| A�� | 44gCO2 �����Ϊ22.4L | |
| B�� | ��״���£�22.4Lˮ�к���NA��ˮ���� | |
| C�� | ���³�ѹ�£�28gN2��������ԭ����ĿΪ2NA | |
| D�� | ���11.2LN2�к���n���������ӣ���ӵ�����һ��Ϊ2n |
| A�� | ����������SO42-Ũ�������� | B�� | ����ͨ��������ͭƬ����пƬ | ||
| C�� | ������O2�ݳ� | D�� | ͭƬ����H2�ݳ� |
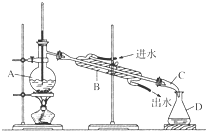 ijͬѧ�������ͼ��ʾװ�ý���ʯ�������ʵ�飬��ش������й����⣺
ijͬѧ�������ͼ��ʾװ�ý���ʯ�������ʵ�飬��ش������й����⣺