��Ŀ����
����Ŀ����1���л���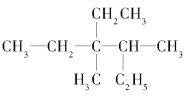 ��ϵͳ������������__��
��ϵͳ������������__��
��2��д��4������2���һ���1����ϩ�Ľṹ��ʽ��__��
��3����������������ͬϵ�����__��
��CH3CH2Cl��CH2=CHCl��CH3CH2CH2Cl��CH2ClCH2Cl��CH3CH2CH2CH3��CH3CH(CH3)2
A.�٢� B.�٢� C.�٢� D.�ݢ�
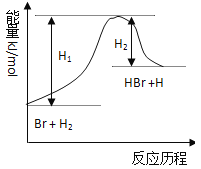
��4��0.1molij����ȼ�գ���ȼ�ղ���ȫ������ʯ�����գ���ʯ����39g�������ķ���ʽΪ___�������ĺ˴Ź���������3���壬��������ܵĽṹ��ʽΪ___��(д������һ�ּ���)
���𰸡�3��4��������3���һ����� ![]() C C6H14 CH3CH2CH2CH2CH2CH3��CH3C(CH3)2CH2CH3
C C6H14 CH3CH2CH2CH2CH2CH3��CH3C(CH3)2CH2CH3
��������
��1����������������ȷ��̼ԭ����Ŀ����̼��Ϊ�������ӿ���֧����һ�˿�ʼ�������ϵ�̼ԭ�ӱ�ţ���дʱ֧���ɼĿ�ʼ����ͬ�Ļ�����Ŀ�ϲ�������й����ţ�����Ϊ���������ŵ��̼�����ӿ���������һ�ʼ��ţ�����ʱҪע���������������ϵ�λ�ã��ݴ˹�����Ը��л���������Ҳ�ɸ���������д�ṹ��ʽ��
��2������ͬϵ���������жϣ�
��3����ʯ�����ؾ��������˶�����̼��ˮ������������ͨʽ�����ɵ�ˮ�Ͷ�����̼�����ʵ�����ʾ������ͨ������ȷ������ʽ��������������ȷ���ṹ��ʽ��
��1��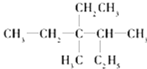 Ϊ�������̼������6��̼ԭ�ӣ�����Ϊ���飬���߶��ǵ�����̼ԭ������֧������֧���϶����ʼ��ţ���3��4��C�ϸ���һ������3��C����һ���һ������л�������Ϊ��3��4��������3���һ����飻
Ϊ�������̼������6��̼ԭ�ӣ�����Ϊ���飬���߶��ǵ�����̼ԭ������֧������֧���϶����ʼ��ţ���3��4��C�ϸ���һ������3��C����һ���һ������л�������Ϊ��3��4��������3���һ����飻
��2��4������2���һ���1����ϩ�����л��������Ϊ��ϩ��������̼̼˫����1��C�ϣ���4��C����1������2��C����1���һ�����ṹ��ʽΪ��![]() ����
����
��3��ͬϵ����ָ�ṹ���ƣ���������һ������CH2ԭ���ŵ��л��ͬϵ���к��еĹ������������Ŀ������ͬ����CH2=CHCl�к���һ��̼̼˫�����������ʶ�û�У�����û����ڻ�Ϊͬϵ����л����CH2ClCH2Cl�к���������ԭ�ӣ��������ʶ�����1�����߲�����ԭ�ӣ�����û����ܻ�Ϊͬϵ����л���ݢ�Ϊ������̼ԭ�Ӹ�����Ϊ4������ͬ���칹�壬������ͬϵ�ֻ�Т�CH3CH2Cl�͢�CH3CH2CH2Cl����ͬϵ�����������߷����ﶼ����1����ԭ�ӣ����Ӽ����1��CH2���ţ����Դ�ѡC��
��4��������ͨʽΪCnH2n+2��0.1mol��������ȫȼ������0.1n molCO2��0.1(n+1) molH2O������Ϊ44��0.1n+18��0.1(n+1)=39�����n=6�������ķ���ʽΪC6H14�����ݺ˴Ź�������ͼ����3����ֵ����3�����͵ĵ�Ч��ԭ�ӣ��������ܵĽṹ��ʽΪCH3CH2CH2CH2CH2CH3��CH3C(CH3)2CH2CH3��