��Ŀ����
2013������������������Ű�ҹ��ж������������У�����β����ȼúβ������ɿ�����Ⱦ��ԭ��֮һ��
(1)����β����������Ҫԭ��Ϊ�� �����ܱ������з����÷�Ӧʱ��c(CO2)���¶�(T)�������ı����(S)��ʱ��(t)�ı仯���ߣ�����ͼ��ʾ��
�����ܱ������з����÷�Ӧʱ��c(CO2)���¶�(T)�������ı����(S)��ʱ��(t)�ı仯���ߣ�����ͼ��ʾ��

�ݴ��жϣ�
�ٸ÷�Ӧ�ġ�H 0(�>������<��)��
����T2�¶��£�0��2s�ڵ�ƽ����Ӧ����v(N2)= ��
�۵��������������һ��ʱ�����������������ѧ��Ӧ���ʡ��������ı����S1>S2������ͼ�л���c(CO2)��T1��S2�����´ﵽƽ������еı仯���ߡ�
�����÷�Ӧ�ں��ݵ��ܱ���ϵ�н��У�����ʾ��ͼ��ȷ����˵���÷�Ӧ���е�t1ʱ�̴ﵽƽ��״̬���� (�����)

(2)ֱ���ŷ�úȼ�ղ������������������صĻ������⡣
��úȼ�ղ����������������������CH4����ԭNOX�������������������Ⱦ��
���磺

д��CH4 (g)����ԭN2O4(g)����N2 (g)��H2O (g)���Ȼ�ѧ����ʽ ��
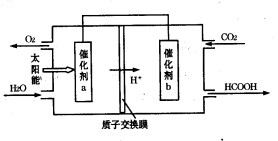
�ڽ�ȼú�����Ķ�����̼�������ã��ɴﵽ��̼�ŷŵ�Ŀ�ġ���ͼ��ͨ���˹�������ã���CO2��H2OΪԭ���Ʊ�HCOOH��O2��ԭ��ʾ��ͼ������b���淢���ĵ缫��ӦʽΪ ��
(14��)(1) ��<.��2�֣� �� 0.025mol/(L��s)��3�֣�.
��  ��2�֣� �� a d��2�֣�
��2�֣� �� a d��2�֣�
(2) �� CH4��g����N2O4��g�� = N2��g����CO2��g����2H2O��g��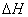 =
-810.1KJ/mol����3�֣�
=
-810.1KJ/mol����3�֣�
�� CO2��2H����2e��=HCOOH.��2�֣�
��������
�����������1���ٸ���ͼ���֪�ڴ����������ͬ�������£�T1���ȴﵽƽ��״̬����˵���¶���T1����T2�����¶ȸ�CO2��Ũ�ȵͣ���˵�������¶�ƽ�����淴Ӧ�����ƶ�����������Ӧ�Ƿ��ȷ�Ӧ������HС��0��
����T2�¶��£�0��2s��CO2��Ũ��������0.1mol/L��������Ũ��������0.05molk/L�����Ե�����ƽ����Ӧ����v(N2)��0.05mol/L��2s��0.025mol/(L��s)��
�۵��������������һ��ʱ�����������������ѧ��Ӧ���ʡ��������ı����S1>S2����Ӧ�ﵽƽ���ʱ����٣���ƽ��״̬�Dz���ģ����������ǣ����𰸣���
����һ�������£������淴Ӧ������Ӧ���ʺ��淴Ӧ�������ʱ������Ϊ0�����������ʵ�Ũ�Ȼ��������ٷ����仯��״̬���ǻ�ѧƽ��״̬������d����˵�������ڸ÷�Ӧ�Ƿ��ȷ�Ӧ�����������¶�ƽ�����淴Ӧ�����ƶ���������Ũ���ǽ��͵ģ�����aͼ�����˵������ȷ�����ݷ���ʽ��֪CO2�͵��������ʵ�����������2:1�ģ�����bͼ����˵����b����ȷ��ƽ��ʱŨ�Ȳ������仯��������֮���Ũ�Ȳ�һ����Ȼ�����ij�ֹ�ϵ������ѡ��c�в���˵�����Ҷ��ߵ�Ũ���DZ仯�ģ�û�дﵽƽ��״̬����ѡad��
(2)�ٸ��ݸ�˹���ɿ�֪��ǰ��ȥ���ߣ����õ���ӦCH4��g����N2O4��g���� N2��g����CO2��g����2H2O��g�������Ը÷�Ӧ�ķ�Ӧ�� ����867kJ/mol��56.9kJ/mol����810.1kJ/mol��
����867kJ/mol��56.9kJ/mol����810.1kJ/mol��
�ڸ���װ��ͼ��֪����������b�缫�ƶ�����˵��b�缫���������õ����ӣ�������ԭ��Ӧ�����Ե缫��Ӧʽ��CO2��2H����2e����HCOOH��
���㣺���鷴Ӧ�ȵ��жϡ����������ƽ��״̬��Ӱ�졢ƽ��״̬���жϡ���Ӧ���ʺͷ�Ӧ�ȵļ����Լ��缫��Ӧʽ����д
�����������Ǹ߿��еij������ͣ��Ѷȴ��ۺ���ǿ����ѧ����Ҫ��ߡ�������ע�ضԻ���֪ʶ���̺�ѵ����ͬʱ�����ض�ѧ�������������ͽ��ⷽ����ָ����ѵ��������������ѧ�������������������ѧ��������û���֪ʶ���ʵ�����������������ѧ����ѧ��������

 �㾦�½̲�ȫ�ܽ��ϵ�д�
�㾦�½̲�ȫ�ܽ��ϵ�д� Сѧ�̲���ȫ���ϵ�д�
Сѧ�̲���ȫ���ϵ�д�
 �����ܱ������з����÷�Ӧʱ��c(CO2)���¶�(T)�������ı����(S)��ʱ��(t)�ı仯���ߣ�����ͼ��ʾ��
�����ܱ������з����÷�Ӧʱ��c(CO2)���¶�(T)�������ı����(S)��ʱ��(t)�ı仯���ߣ�����ͼ��ʾ�� 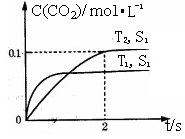
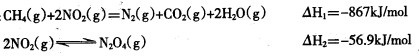





 2CO2(g)+
N2(g)����H��0
2CO2(g)+
N2(g)����H��0

 N2O4(g) ��H����56.9 kJ/mol
N2O4(g) ��H����56.9 kJ/mol