��Ŀ����
���������ھ�������������ˮ��������������ҵ�������Ư����
��1���������������������ҿ������������е��ʷ�Ӧ���磺6Ag��s����O3��g��=3Ag2O��s������H����235.8 kJ��mol��1����֪2Ag2O��s��=4Ag��s����O2��g�� ��H����62.2 kJ��mol��1�������·�Ӧ2O3��g��=3O2��g���Ħ�H��________��
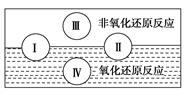
��2����ƽ���淴Ӧ�Ļ�ѧ����ʽ���������ʵĻ�ѧ������������Ӧ�ķ����ڣ���
��3����ѧ��P.Tatapudi��������ʹ�������������µ��ˮ�ķ����Ƶó�����������������Χ��ˮ�в������缫��ӦʽΪ3H2O��6e��=O3����6H�������������ܽ���ˮ�е��������ɹ������⣬��缫��ӦʽΪ_______________________��
��4�������г����ļ�ⷽ���ǽ���������ͨ������KI������Һ������Һ����ɫ����˵�������к���O3����֪O3��KI��Һ��Ӧ�������ֵ��ʣ���÷�Ӧ�����ӷ���ʽΪ_____________________________________________________��
��1����285.0 kJ��mol��1
��2��2��3��1��1��3��1
��3��3O2��6H����6e��=3H2O2����O2��2H����2e��=H2O2��
��4��2I����O3��H2O=2OH����I2��O2
����

��������(H2O2)��һ����ɫ��Һ��,����ˮ��Һ�׳�˫��ˮ,��������,����������������ɱ������Ư���ȡ�
(1)����˵����ȷ���� ��
| A��������������м��м��Լ����зǼ��Լ� |
| B��H2O2��H2O��Ϊͬ�������� |
| C��34 g H2O2�к��е���������ΪNA |
| D��ʵ���ҿ������ù���������ȡ���� |
(3)��H2O2��Һ��������FeCl2��Һ��,��Һ��dz��ɫ��Ϊ�ػ�ɫ,д���÷�Ӧ�����ӷ���ʽ: ��
(4)ij����ҵ��ˮ�к���һ��������,Ϊ�˳�ȥ����,������H2O2�����ȼ�,д���÷�Ӧ�Ļ�ѧ����ʽ: ��
 Cu��____CuCl2��N2����____H2O��
Cu��____CuCl2��N2����____H2O�� ��
�� Cr2O72-����ɫ����H2O��
Cr2O72-����ɫ����H2O�� Cr3����aq����3OH����aq���������£�Cr��OH��3���ܶȻ�Ksp��10��32����c��Cr3��������10��5 mol��L��1ʱ����Ϊc��Cr3�����Ѿ���ȫ�������ֽ��ڢ۲���Һ��pH����4����ͨ������˵��Cr3���Ƿ������ȫ����д��������̣���____________________________________________________________________________��
Cr3����aq����3OH����aq���������£�Cr��OH��3���ܶȻ�Ksp��10��32����c��Cr3��������10��5 mol��L��1ʱ����Ϊc��Cr3�����Ѿ���ȫ�������ֽ��ڢ۲���Һ��pH����4����ͨ������˵��Cr3���Ƿ������ȫ����д��������̣���____________________________________________________________________________��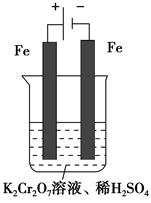

 CeO2��8OH + 8_____����CeO2��8OH
CeO2��8OH + 8_____����CeO2��8OH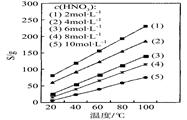

 2NO2 (g) ���� һ���¶��£���Ӧ�����ʱ�Ϊ��H���ֽ�1 mol N2O4 ����һ��ѹ�ܱ������У�����ʾ��ͼ��ȷ����˵����Ӧ�ﵽƽ��״̬����________��
2NO2 (g) ���� һ���¶��£���Ӧ�����ʱ�Ϊ��H���ֽ�1 mol N2O4 ����һ��ѹ�ܱ������У�����ʾ��ͼ��ȷ����˵����Ӧ�ﵽƽ��״̬����________��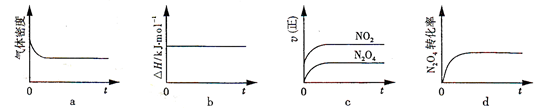
 ��I
��I