��Ŀ����
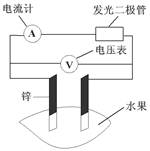
����ijǦ���ص�����������DZ�ĥ��������ͼװ�����ʵ�飬ʶ�����Ǧ���ص���������
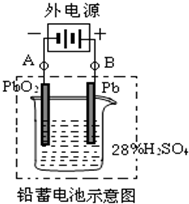
��1����A��E��B��F����B�缫����______����ӦʽΪ______����˵��FΪ������
��2��Ǧ���ع���ʱ���ŵ磩����E���ڵ缫�ĵ缫��ӦʽΪ��______�����ʱ�ü�����ӵ�Դ��______��������
��3������0.2mol���ӷ���ת�ƣ����������ĵ�PbO2�����ʵ�����______��
�������ü״���CH3OH�����������ȼ�ϵ�أ��������ҺΪ���ԣ���ع���ʱ���为������ʽ��д���ӷ���ʽ��Ϊ______��
����һ�ֻ�������ȼ�ϵ��ԭ����Ƶľƾ�����ǣ������ϵķ�ӦΪCH3CH2OH+H2O��CH3COOH+4H++4e-�������ķ�Ӧʽ��______����ط�Ӧ���ܷ���ʽΪ��______��

��1����A��E��B��F����B�缫����______����ӦʽΪ______����˵��FΪ������
��2��Ǧ���ع���ʱ���ŵ磩����E���ڵ缫�ĵ缫��ӦʽΪ��______�����ʱ�ü�����ӵ�Դ��______��������
��3������0.2mol���ӷ���ת�ƣ����������ĵ�PbO2�����ʵ�����______��
�������ü״���CH3OH�����������ȼ�ϵ�أ��������ҺΪ���ԣ���ع���ʱ���为������ʽ��д���ӷ���ʽ��Ϊ______��
����һ�ֻ�������ȼ�ϵ��ԭ����Ƶľƾ�����ǣ������ϵķ�ӦΪCH3CH2OH+H2O��CH3COOH+4H++4e-�������ķ�Ӧʽ��______����ط�Ӧ���ܷ���ʽΪ��______��

����1����ԭ�������������Ϊ���ص�����������BΪ������������������ʧ���ӣ���������������B�缫���ֻ���ɫ���壬��缫��ӦʽΪ2Cl--2e-=Cl2����
�ʴ�Ϊ������ɫ�����������2Cl--2e-=Cl2����
��2��Ǧ���ع���ʱ����ΪPbʧ���ӣ�����缫��ӦʽΪ��Pb+SO42--2e-=PbSO4�����ʱ���صĸ�������ӵ�Դ�ĸ���������
�ʴ�Ϊ��Pb+SO42--2e-=PbSO4������
��3��������PbO2��PbSO4��PbԪ�ش�+4���͵�+2��ת��2�����ӣ���������0.2mol���ӷ���ת�ƣ����������ĵ�PbO2�����ʵ�����0.1mol��
�ʴ�Ϊ��0.1mol��
��II�����������£������ϼ״�ʧ��������̼������ӣ�����缫��ӦʽΪ��CH3OH+8OH--6e-=CO32-+6H2O���ʴ�Ϊ��CH3OH+8OH--6e-=CO32-+6H2O��
��III������ȼ�ϵ���������������õ�������ˮ����缫��ӦʽΪ��O2+4H++4e-=2H2O�������ĵ缫��Ӧʽ������ĵ缫��Ӧʽ��Ӽ����ܷ�Ӧ�����Ե�ط�Ӧ���ܷ���ʽΪ��CH3CH2OH+O2=CH3COOH+H2O���ʴ�Ϊ��O2+4H++4e-=2H2O��CH3CH2OH+O2=CH3COOH+H2O��
�ʴ�Ϊ������ɫ�����������2Cl--2e-=Cl2����
��2��Ǧ���ع���ʱ����ΪPbʧ���ӣ�����缫��ӦʽΪ��Pb+SO42--2e-=PbSO4�����ʱ���صĸ�������ӵ�Դ�ĸ���������
�ʴ�Ϊ��Pb+SO42--2e-=PbSO4������
��3��������PbO2��PbSO4��PbԪ�ش�+4���͵�+2��ת��2�����ӣ���������0.2mol���ӷ���ת�ƣ����������ĵ�PbO2�����ʵ�����0.1mol��
�ʴ�Ϊ��0.1mol��
��II�����������£������ϼ״�ʧ��������̼������ӣ�����缫��ӦʽΪ��CH3OH+8OH--6e-=CO32-+6H2O���ʴ�Ϊ��CH3OH+8OH--6e-=CO32-+6H2O��
��III������ȼ�ϵ���������������õ�������ˮ����缫��ӦʽΪ��O2+4H++4e-=2H2O�������ĵ缫��Ӧʽ������ĵ缫��Ӧʽ��Ӽ����ܷ�Ӧ�����Ե�ط�Ӧ���ܷ���ʽΪ��CH3CH2OH+O2=CH3COOH+H2O���ʴ�Ϊ��O2+4H++4e-=2H2O��CH3CH2OH+O2=CH3COOH+H2O��

��ϰ��ϵ�д�
 �ݾ�ѵ������ϵ�д�
�ݾ�ѵ������ϵ�д� С����ȫ�ܼ��ϵ�д�
С����ȫ�ܼ��ϵ�д�
�����Ŀ


 ��
��

 2SO3(g)��ѹ���������ʹѹǿ���������������䣩����SO2��ת��������ƽ�ⳣ��KҲ����
2SO3(g)��ѹ���������ʹѹǿ���������������䣩����SO2��ת��������ƽ�ⳣ��KҲ���� ��ֵ����
��ֵ���� CO(g)+H2(g)����������жϸ÷�Ӧ�ﵽƽ��״̬�ı�־�� ��(����ĸ)
CO(g)+H2(g)����������жϸ÷�Ӧ�ﵽƽ��״̬�ı�־�� ��(����ĸ) HCOOCH3(g) ��H=��29.1 kJ��mol-1
HCOOCH3(g) ��H=��29.1 kJ��mol-1