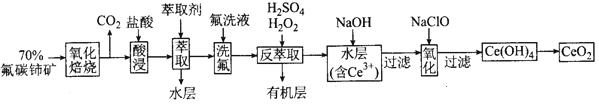
��Ŀ����
����Ŀ������������һ���ķ����,��ijЩ������ˮ��Ӧ������з����ͼʾ��
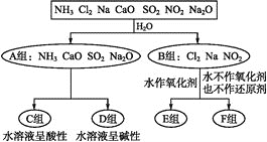
���������ѧ��֪ʶ,��Ҫ�����:
(1)������һ�������(�ֳ�A��B�������)��____��
(2)F�������ʳ���Cl2���____(�ѧʽ)��
(3)A���е�CaO��������ʳƷ��װ���еĸ����,CaO��������������Ϊ____(��������)��
�ٽ���������ڼ���������ۼ�ܼ��Ը����
CaO�����������������____________(�û�ѧ����ʽ��ʾ)��
(4)D����NH3��ˮ��Һ��������,�õ��뷽��ʽ��ʾ��������Ե�ԭ��:________��
(5)��Al3+�Ʊ�Al(OH)3,��ò�ѡ��D���е�NaOH��Һ,˵������:___________��
���𰸡���1���Ƿ���������ԭ��Ӧ ��2��NO2
��3���٢ڢ� CaO+H2OCa��OH��2
��4��NH3��H2O![]() N
N![]() +OH-
+OH-
��5������NaOH��Һ���ܽ����ɵ�Al��OH��3����
��������
��1������A��B����������ˮ�ķ�Ӧ�����֪��һ����������Ƿ���������ԭ��Ӧ��
��2��Cl2��NO2��ˮ��Ӧʱ,ˮ�Ȳ���������Ҳ������ԭ����
��3��CaO����ˮ��Ӧ,����ˮ����,���٢ڢ���ȷ��
��4��D����NH3��ˮ��Һ�������Ե�ԭ����:NH3��H2O��������:NH3��H2O![]() N
N![]() +OH-,����OH-��
+OH-,����OH-��
��5����Al3+�Ʊ�Al��OH��3,��ò�ѡ��NaOH��Һ,��Ϊ����NaOH��Һ���ܽ����ɵ�Al��OH��3������