��Ŀ����
��6�֣���������ЧӦ����Դ��ȱ�����⣬��ν��ʹ����е�CO2���������Կ������ã������˸������ձ����ӡ�Ŀǰ��ҵ����һ�ַ�������CO2����ȼ�ϼ״���
��1����250C��101KPaʱ��3.2�˼״���CH30H����ȫȼ������CO2��Һ̬ˮʱ����72.576kJ�����ܱ�ʾ�״�ȼ�յ��Ȼ�ѧ����ʽΪ___________________��
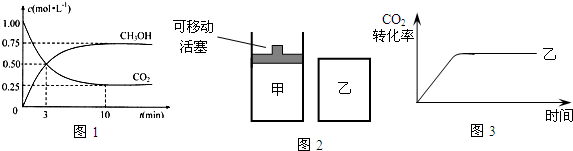
��2��Ϊ̽����Ӧԭ�����ֽ�������ʵ�飬�����Ϊ2 L���ܱ������У�����2mol CO2��6mol H2��һ�������·�����Ӧ��CO2(g)��3H2(g)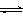 CH3OH(g)��H2O(g) ��H����49kJ/mol�����CO2��H2O (g)��Ũ����ʱ��仯��ͼ��ʾ��
CH3OH(g)��H2O(g) ��H����49kJ/mol�����CO2��H2O (g)��Ũ����ʱ��仯��ͼ��ʾ��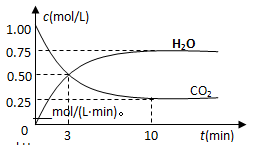
�ٴӷ�Ӧ��ʼ��ƽ�⣬������ƽ����Ӧ����v(H2)��__________mol/(L��min)��
�����д�ʩ����ʹn(H2O)��n(CO2)�������________��
A�������¶� B���ٳ���3mol H2
C����CH3OH (g)����ϵ�з��� D������He(g)��ʹ��ϵѹǿ����
��6�֣���1��CH3OH(l)+3/2O2(g)=CO2(g)+2H2O(l) ��H=-725.76kJ��mol-1
��2����0.225 mol/(L��min)����B C ��ÿ��2�֣�
����

��ϰ��ϵ�д�
 �ǻ�С��ϰϵ�д�
�ǻ�С��ϰϵ�д�
�����Ŀ
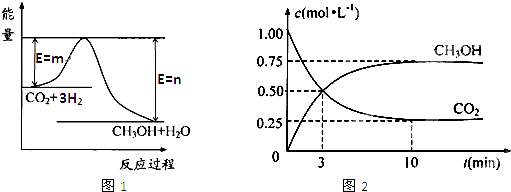
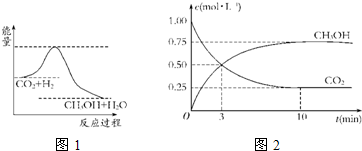
 ������CO2����ȼ�ϼ״���һ�������·�����Ӧ��CO2��g��+3H2��g��
������CO2����ȼ�ϼ״���һ�������·�����Ӧ��CO2��g��+3H2��g�� CH3OH��g��+H2O��g����������ͼ��ʾ�÷�Ӧ���й�������������λΪkJ?mol-1���ı仯��
CH3OH��g��+H2O��g����������ͼ��ʾ�÷�Ӧ���й�������������λΪkJ?mol-1���ı仯��