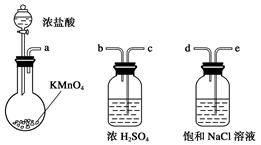
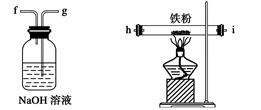
��Ŀ����
��֪�Ҵ����Ժ��Ȼ��Ʒ�Ӧ��������ˮ��CaCl2��6C2H5OH���йص��л��Լ��ķе����£�CH3COOC2H5Ϊ77.1�棻C2H5OHΪ78.3�棻C2H5OC2H5�����ѣ�Ϊ34.5�棻CH3COOHΪ118�档ʵ���Һϳ����������ֲ�Ʒ�IJ������£���������ƿ�ڽ��������Ҵ�������Ũ�����ϣ�Ȼ��Һ©���ߵμӴ��ᣬ���������������ʵ��ɵõ������Ҵ������ѡ������ˮ�����������ֲ�Ʒ��
��1����Ӧ�м�����Ҵ��ǹ����ģ���Ŀ���� ��
��2���ߵμӴ��ᣬ���������Ŀ���� ��
���ֲ�Ʒ�پ����в��辫�ƣ�
��3��Ϊ��ȥ���еĴ��ᣬ�����Ʒ�м��� ������ĸ����
A.��ˮ�Ҵ� B.̼���Ʒ�ĩ C.��ˮ������
��4���������м��뱥���Ȼ�����Һ�������룬��Ŀ���� ��
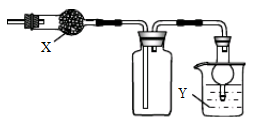
��5��Ȼ���������м�����ˮ����ͭ������Ŀ���� ��������������������Һ�������һ���������ƿ�ڣ���������ȥ�ͷе���֣��ռ��е���76��~78��֮�����ּ��ô���������������
��1����Ӧ�м�����Ҵ��ǹ����ģ���Ŀ���� ��
��2���ߵμӴ��ᣬ���������Ŀ���� ��
���ֲ�Ʒ�پ����в��辫�ƣ�
��3��Ϊ��ȥ���еĴ��ᣬ�����Ʒ�м��� ������ĸ����
A.��ˮ�Ҵ� B.̼���Ʒ�ĩ C.��ˮ������
��4���������м��뱥���Ȼ�����Һ�������룬��Ŀ���� ��
��5��Ȼ���������м�����ˮ����ͭ������Ŀ���� ��������������������Һ�������һ���������ƿ�ڣ���������ȥ�ͷе���֣��ռ��е���76��~78��֮�����ּ��ô���������������
��1������Ӧ���Ҵ���Ũ�ȣ������ڷ�Ӧ���������������ķ������
��2�����Ӵ���Ũ�ȣ���С����������������Ũ�ȣ�������������Ӧ���������������ķ������
��3��B
��4����ȥ�ֲ�Ʒ�е��Ҵ�
��5����ȥ�ֲ�Ʒ�е�ˮ
��2�����Ӵ���Ũ�ȣ���С����������������Ũ�ȣ�������������Ӧ���������������ķ������
��3��B
��4����ȥ�ֲ�Ʒ�е��Ҵ�
��5����ȥ�ֲ�Ʒ�е�ˮ
�����������1����ȡ���������ķ�Ӧ�ǿ��淴Ӧ������Ӧ���Ҵ���Ũ�ȣ������ڷ�Ӧ���������������ķ�����У�
��2�������������Ҵ������ѵķе�ϵͣ��Ӵ��������Ŀ��ʹ��Ʒ��������С����������Ũ�ȣ�ͬʱ��������Ũ�ȣ�ʹƽ��������У�
��3����������������ˮ��������̼���Ʒ�Ӧ���ɴ����ơ�ˮ��������̼���壬��������ˮ���������²㣬������ˮ�������������ϲ㣬��Һ��ʹ�������룬��˳�ȥ��Ʒ�еĴ��ᣬӦѡ��̼������Һ����ѡB��
��4���ֲ�Ʒ���Ժ����Ҵ������ѡ�ˮ�������Ҵ����Ժ��Ȼ��Ʒ�Ӧ��������ˮ��CaCl2��6C2H5OH�����Լ��뱥�͵��Ȼ�����Һ��Ŀ���dz�ȥ�ֲ�Ʒ�е��Ҵ���
��5����ˮ����ͭ��ˮ���������ɫ��CuSO4��5H2O�����Լ�����ˮ����ͭ������Ŀ���dz�ȥ�ֲ�Ʒ�е�ˮ��Ȼ������ɵ�����������

��ϰ��ϵ�д�
�����Ŀ
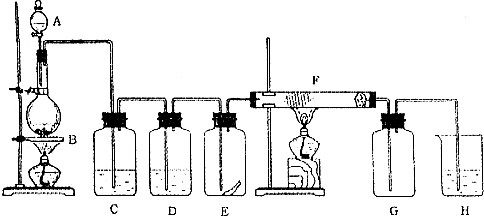


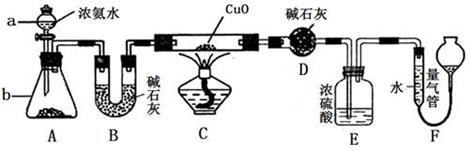
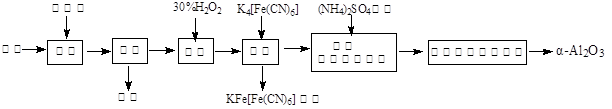
 2Al2O3 + 2NH3��+ N2��+ 5SO3��+ 3SO2��+ 53H2O,������������ͨ��ͼ9��ʾ��װ�á�
2Al2O3 + 2NH3��+ N2��+ 5SO3��+ 3SO2��+ 53H2O,������������ͨ��ͼ9��ʾ��װ�á�