��Ŀ����
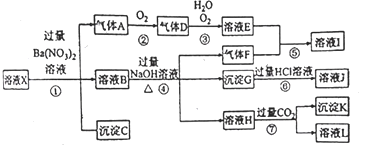
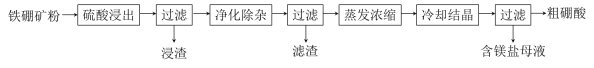
����Ŀ�����仯�����ڹ�ҵ����������;�����������Ҫ�ɷ�ΪMg2B2O5��H2O��Fe3O4����������Fe2O3��FeO��CaO��Al2O3��SiO2��)Ϊԭ���Ʊ�����(H3BO3)�Ĺ���������ͼ��ʾ��
��֪������Ϊ��ĩ״���壬������ˮ�����ȵ�һ���¶ȿɷֽ�Ϊ��ˮ�
�ش��������⣺
��1��д��Mg2B2O5��H2O�����ᷴӦ�Ļ�ѧ����ʽ___��Ϊ��߽������ʣ����ʵ���������Ũ��Ũ���⣬���ɲ�ȡ�Ĵ�ʩ�У�д��������___��
��2�����������л�ʣ��������ǣ�___��д��ѧʽ����
��3�����������������ȼ�H2O2��Һ��������___��Ȼ���ٵ�����Һ��pHԼΪ5��Ŀ����___��
��4�������������е���Ҫ������___�������ƣ���
��5����������������ɾ������������������֡�������ͽ���þΪԭ�Ͽ��Ʊ��������û�ѧ����ʽ��ʾ�Ʊ�����___��
���𰸡�Mg2B2O5��H2O+2H2SO4![]() 2MgSO4+2H3BO3 ��С��������������߷�Ӧ�¶� SiO2��CaSO4 ��Fe2+����ΪFe3+ ʹAl3+��Fe3+�γ������������ȥ ����ˮ������þ 2H3BO3
2MgSO4+2H3BO3 ��С��������������߷�Ӧ�¶� SiO2��CaSO4 ��Fe2+����ΪFe3+ ʹAl3+��Fe3+�γ������������ȥ ����ˮ������þ 2H3BO3![]() B2O3+3H2O��B2O3+3Mg
B2O3+3H2O��B2O3+3Mg![]() 3MgO+2B
3MgO+2B
��������
�������(��Ҫ�ɷ�ΪMg2B2O5��H2O��Fe3O4����������Fe2O3��FeO��CaO��Al2O3��SiO2��)Ϊԭ���Ʊ�����(H3BO3)�������̿�֪���������ܽ⣬Fe3O4��SiO2���ܣ�CaOת��Ϊ����ˮ��CaSO4��Fe3O4���д��ԣ����Բ��������������룬����1�ijɷ�ΪSiO2��CaSO4��
���������������ȼ�H2O2��Һ����Fe 2+ת��ΪFe 3+��������Һ��pHԼΪ5��ʹFe3����Al3����ת��Ϊ������������ΪAl(OH)3��Fe(OH)3��Ȼ������Ũ������ȴ�ᾧ�����˷����H3BO3��
(1)Mg2B2O5��H2O�����ᷴӦ��������þ�����ᣬ��ѧ����ʽ Mg2B2O5��H2O+2H2SO4![]() 2MgSO4+2H3BO3��
2MgSO4+2H3BO3��
Ϊ��߽������ʣ����ʵ���������Ũ��Ũ���⣬���ɲ�ȡ�Ĵ�ʩ�У���С��������������߷�Ӧ�¶ȣ�
(2)����Fe3O4�Ĵ��ԣ��ɽ�������������з��룬���������л�ʣ���������SiO2��CaSO4��
(3)���������������ȼ�H2O2��Һ�������ǽ�Fe2+����ΪFe3+ ��Ȼ���ٵ�����Һ��pHԼΪ5��Ŀ����ʹAl3+��Fe3+�γ������������ȥ��
(4)���ݷ���ʽMg2B2O5��H2O+2H2SO4![]() 2MgSO4+2H3BO3����Ӧ�л���������þ�������������е���Ҫ������(��ˮ)����þ��
2MgSO4+2H3BO3����Ӧ�л���������þ�������������е���Ҫ������(��ˮ)����þ��
(5)����ͽ���þΪԭ�Ͽ��Ʊ��������������ȷֽ⣬��������������þ����ԭ����ѧ����ʽ��2H3BO3![]() B2O3+3H2O��B2O3+3Mg
B2O3+3H2O��B2O3+3Mg![]() 3MgO+2B��
3MgO+2B��

����Ŀ����֪ X��Y��Z��Q��R��E����ǰ������Ԫ���У�ԭ������X��Y��Z��Q��R��E����ṹ��������Ϣ�����
Ԫ�� | �ṹ��������Ϣ |
X | ԭ�ӵ�L����s����������p������ |
Y | ԭ�Ӻ����L����3��δ�ɶԵ��� |
Z | �ؿ��к�������Ԫ�� |
Q | ���ʳ��³�ѹ�������壬ԭ�ӵ�M������1��δ�ɶԵ�p���� |
R | �˵������Y��Q�ĺ˵����֮�� |
E | N�ܲ���ֻ��һ�����ӣ�K��L��M����������� |
�������Ϣ�ش��й����⣺
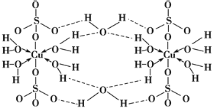
(1)д��Ԫ��Y��ԭ�Ӻ���۵����Ų�ͼ��___________��X��һ���⻯����Է�������Ϊ26��������е������������ļ���֮��Ϊ______��
(2)X��Y��Z����Ԫ�صĵ�һ�������ɸߵ��͵�����Ϊ(дԪ�ط���)_____��
(3)X��ZԪ�طֱ�����Ԫ���γɵ���������У��е�ϸߵ�Ϊ(д��ѧʽ)______��ԭ����________��
(4)XZ��Y2���ڵȵ����壬д��������XZ�Ľṹʽ��_____��
(5)R��һ�������Ļ�ѧʽΪRCl36H2O����֪0.01mol RCl36H2O��ˮ��Һ���ù�����������Һ����������0.02mol AgCl��������������������____��
A��[R(H2O)6]Cl3 B��[R(H2O)5Cl]Cl2H2O
C��[R(H2O)4Cl2]Cl2H2O D��[R(H2O)3Cl3]3H2O
(6)������ESO4��ˮ��Һ����ε��백ˮ��������ɫ��������Ӧ�����ӷ���ʽΪ��__�������μӰ�ˮ�������������ܽ⣬�õ�����ɫ��Һ��д����Ӧ�����ӷ���ʽΪ��__��