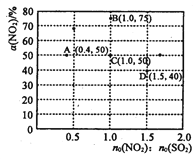
��Ŀ����
����Ŀ���״�ȼ�ϵ�����С���ྻ���������������ȸߣ����ڱ�ЯʽͨѶ�豸������������Ӧ�á�ij�״�ȼ�ϵ�ص��ܷ�Ӧʽ2CH4O + 3O2= 2CO2+ 4H2O����ͼ�Ǹ�ȼ�ϵ�ص�ʾ��ͼ������˵����ȷ����
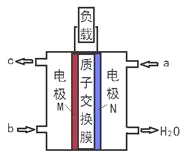
A. a�Ǽ״�ȼ�ϡ�b������
B. ��ת��6mole-ʱ������33.6LO2
C. ������Ӧ��CH4O - 6e-+ H2O = CO2��+ 6H+
D. ���Ӵ�N�缫����������Ĥ����M�缫��
���𰸡�C
����������ȼ�ϵ���У�ͨ��ȼ�ϵĵ缫�Ǹ�����ͨ���������ĵ缫������������ͼƬ֪���������Һ�����ԣ�N��������ˮ����aΪ������NΪ������MΪ������������ӦʽΪCH40-6e-+H20=C02��+6H+��������ӦʽΪ02+4H++4e-=H20��b�Ǽ״���c�Ƕ�����̼�����ӴӸ���ͨ�����ӽ���Ĥ��������A��ͨ�����Ϸ���֪��a��������b�Ǽ״�����A����B��û�б��������״̬����֪�����Ħ����������������ʵ�������������������B��ȷ��C�������ϼ״�ʧ���ӷ���������Ӧ���缫��ӦʽΪCH4O-6e-+H2O=CO2��+6H+����C��ȷ��D��M�Ǹ�����N�����������Ӵ�M�缫����������Ĥ������N�缫�ƶ�����D����ѡC��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ��Ϊ�˺������û�ѧ�ܣ�ȷ����ȫ���������������Ҫ��ֿ��ǻ�ѧ��Ӧ�ķ�Ӧ�ȣ�����ȡ��Ӧ��ʩ����ѧ��Ӧ�ķ�Ӧ��ͨ����ʵ����вⶨ��Ҳ�ɽ����������㡣
��1��ʵ���ã�5g�״���CH3OH��Һ���������г��ȼ�����ɶ�����̼�����Һ̬ˮʱ�ͷų�113.5kJ�����������ʾ�״���ȼ���ȵ��Ȼ�ѧ����ʽΪ�� ��
��2���������������Ȼ�ѧ����ʽ����a b����������������������������
H2(g)+ 1/2O2(g)��H2O(g) ��H1��a kJ��mol-1
H2(g)+ 1/2O2(g)��H2O(l) ��H2��b kJ��mol-1
��3����1mol��̬������ij�ֹ��ۼ���Ҫ���յ������м��ܡ��ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̡��ڻ�ѧ��Ӧ�����У���ѧ����Ҫ�����������γɻ�ѧ���ֻ��ͷ�������
��ѧ�� | H��H | N��H | N��N |
����/kJ��mol��1 | 436 | 391 | 945 |
��֪��ӦN2(g)��3H2(g)![]() 2NH3(g) ��H��a kJ��mol��1���Ը��ݱ������м������ݹ���a��ֵ��_______________(ע����+����������)��
2NH3(g) ��H��a kJ��mol��1���Ը��ݱ������м������ݹ���a��ֵ��_______________(ע����+����������)��
��4�����ݸ�˹���ɿ��Զ�ijЩ����ͨ��ʵ��ֱ�Ӳⶨ�Ļ�ѧ��Ӧ�ķ�Ӧ�Ƚ������㡣����ˮú���ϳɶ����ѵ�������Ӧ���£�
�� 2H2(g) + CO(g)![]() CH3OH(g)����H ����90.8 kJ��mol��1
CH3OH(g)����H ����90.8 kJ��mol��1
�� 2CH3OH(g)![]() CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1
CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1
�� CO(g) + H2O(g)![]() CO2(g) + H2(g)����H����41.3 kJ��mol��1
CO2(g) + H2(g)����H����41.3 kJ��mol��1
�ܷ�Ӧ��3H2(g) + 3CO(g)![]() CH3OCH3(g) + CO2(g)����H�� ��
CH3OCH3(g) + CO2(g)����H�� ��
����Ŀ������������һ�ֹ�ҵ�Σ������ʳ�ηdz����ƣ����Խ�ǿ��
��.���飺��Ksp(AgNO2)=2��10��8��Ksp(AgCl)=1.8��10��10����Ka(HNO2)=5.1��10��4���������ķ�������NaNO2��NaCl���ֹ���______________________
��. ijС��ͬѧ������װ�ã���ȥ�г��������Ʊ���������
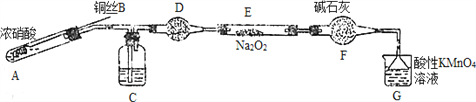
��֪����2NO��Na2O2=2NaNO2�� �����������£�NO��NO2������MnO4����Ӧ����NO3����Mn2����
��1��ʹ��ͭ˿���ŵ���________________________��
��2��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ_____________________________��
װ��C��ʢ�ŵ�ҩƷ��_________��������ĸ���ţ�
A��Ũ���� B��NaOH��Һ C��ˮ D�����Ȼ�̼
��3����С���ȡ5.000g��ȡ����Ʒ����ˮ���250ml��Һ��ȡ25.00ml��Һ����ƿ�У�
��0.1000mol��L��1����KMnO4��Һ���еζ���ʵ�������������±���ʾ��
����� | 1 | 2 | 3 | 4 |
����KMnO4��Һ���/mL | 20.90 | 20.12 | 20.00 | 19.88 |
�ٵ�һ��ʵ�����ݳ����쳣����������쳣��ԭ�������_________������ĸ���ţ���
a����ƿϴ����δ����
b����ʽ�ζ���������ˮϴ����δ�ñ�Һ��ϴ
c���ζ��յ�ʱ���Ӷ���
������KMnO4��Һ�ζ�����������Һ�����ӷ���ʽΪ___________________��
�۸���Ʒ���������Ƶ���������Ϊ______________��