��Ŀ����
����Ŀ��ԭ�������������������Ԫ��A��B��C��D�ֱ��ڵ�һ���������ڣ���Ȼ���д��ڶ���A�Ļ����Bԭ�Ӻ��������6�ֲ�ͬ���˶�״̬��B��C���γ����������η��ӣ�D�Ļ�̬ԭ�ӵ������ܲ�ֻ��һ�����ӣ������ܲ���ѳ������ӡ���ش��������⣺
��1��������Ԫ���е縺������Ԫ�أ����̬ԭ�ӵļ۵����Ų�ͼΪ____________________����һ��������С��Ԫ����__________(��Ԫ�ط���)��
��2��C���������ǰ����Ԫ�طֱ���A�γɵĻ�����е��ɸߵ��͵�˳����___________(�ѧʽ)��������˵ݱ���ɵ�ԭ����______________________________��
��3��BԪ�ؿ��γɶ��ֵ��ʣ�һ�־���ṹ��ͼһ��ʾ����ԭ�ӵ��ӻ�����Ϊ__________����һ�ֵľ�����ͼ����ʾ���þ����Ŀռ�������Ϊ__________�����˾����е��ⳤΪ356.6 pm����˾������ܶ�Ϊ__________g��cm3(������λ��Ч����)��(![]() =1.732)
=1.732)
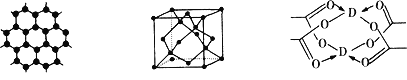
ͼһ ͼ�� ͼ��
��4��DԪ���γɵĵ��ʣ��侧��Ķѻ�ģ��Ϊ__________��D�Ĵ����ξ���ֲ��ṹ��ͼ�����þ����к��еĻ�ѧ����__________ (��ѡ�����)��
�ټ��Լ� �ڷǼ��Լ� ����λ�� �ܽ�����
��5����D����������Һ�еμӹ�����ˮ���۲쵽��������______________________________��
��д���������̵����ӷ���ʽ��__________________________________________________��
���𰸡�![]() Cu HF��HI��HBr��HCl HF����֮���γ����ʹ���۷е�ϸߣ�HI��HBr��HCl����֮��ֻ�з��»�������Է�������Խ���»���Խ�� sp2 34% 3.5 �����������ܶѻ� �٢ڢ� �����γ���ɫ�����������μӰ�ˮ�������ܽ⣬�õ�����ɫ������Һ Cu2++2NH3��H2O==Cu(OH)2��+NH4+��Cu(OH)2+4NH3==[Cu(NH3)4]2++2OH
Cu HF��HI��HBr��HCl HF����֮���γ����ʹ���۷е�ϸߣ�HI��HBr��HCl����֮��ֻ�з��»�������Է�������Խ���»���Խ�� sp2 34% 3.5 �����������ܶѻ� �٢ڢ� �����γ���ɫ�����������μӰ�ˮ�������ܽ⣬�õ�����ɫ������Һ Cu2++2NH3��H2O==Cu(OH)2��+NH4+��Cu(OH)2+4NH3==[Cu(NH3)4]2++2OH
��������
ԭ�������������������Ԫ��A��B��C��D�ֱ��ڵ�һ���������ڣ���Ȼ���д��ڶ���A�Ļ������AΪ��Ԫ�أ�Bԭ�Ӻ��������6�ֲ�ͬ���˶�״̬����������6�����ӣ���BΪ̼Ԫ�أ�D�Ļ�̬ԭ�ӵ������ܲ�ֻ��һ�����ӣ������ܲ���ѳ������ӣ�Dԭ����Χ�����Ų�Ϊ3d104s1����DΪͭԪ�أ����ԭ��������֪��Cֻ�ܴ��ڵ������ڣ�B��C���γ����������ͷ��ӣ���BΪ��Ԫ�ء�
��1������Ԫ���е縺��������Cl�����̬ԭ�ӵļ۵�����Ϊ7��������Խǿ����һ������ԽС��
��2��HF���Ӽ����������е���ߣ�����±����������Է���������������е����ߣ�
��3��ͼһΪƽ��ṹ�������״�ṹ��̼̼������Ϊ120����ÿ��̼ԭ�Ӷ������3��̼ԭ�ӣ�̼ԭ�Ӳ�ȡsp2�ӻ������㾧����̼ԭ��������뾧���������������þ�����ÿ��Cԭ������Χ4��Cԭ���γ���������ṹ��ȷ�������ⳤ��Cԭ�Ӱ뾶��ϵ�����ݾ�̯�����㾧����Cԭ����Ŀ���������㾧���������ٸ��ݾ����ܶ�=����/������㣻
��4������CuΪ�����������ܶѻ������ͼ������ͭ����ľֲ��ṹ��ȷ���侧���к��м��Լ����Ǽ��Լ�����λ����
��5������ͭ��Һ�м��백ˮ�����������ͭ��ɫ�����������μӰ�ˮ�������ܽ⣬�õ��İ���ͭ�����ӣ���ҺΪ����ɫ������Һ��
ԭ�������������������Ԫ��A��B��C��D�ֱ��ڵ�һ���������ڣ���Ȼ���д��ڶ���A�Ļ������AΪ��Ԫ�أ�Bԭ�Ӻ��������6�ֲ�ͬ���˶�״̬����������6�����ӣ���BΪ̼Ԫ�أ�D�Ļ�̬ԭ�ӵ������ܲ�ֻ��һ�����ӣ������ܲ���ѳ������ӣ�Dԭ����Χ�����Ų�Ϊ3d104s1����DΪͭԪ�أ����ԭ��������֪��Cֻ�ܴ��ڵ������ڣ�B��C���γ����������ͷ��ӣ���BΪ��Ԫ�ء�
��1������Ԫ���е縺��������Cl�����̬ԭ�ӵļ۵�����Ϊ7�����̬ԭ�ӵļ۵����Ų�ͼΪ![]() ����Ԫ����Cu�Ľ�����Խǿ����Cu�ĵ�һ������ԽС��
����Ԫ����Cu�Ľ�����Խǿ����Cu�ĵ�һ������ԽС��
�ʴ�Ϊ��![]() ��Cu��
��Cu��
��2��HF����֮���γ����ʹ���۷е�ϸߣ�HI��HBr��HCl����֮��ֻ�з��»�������Է�������Խ���»���Խ�е�Խ�ߣ����е��ɸߵ��͵�˳����HF��HI��HBr��HCl��
�ʴ�Ϊ��HF��HI��HBr��HCl��HF����֮���γ����ʹ���۷е�ϸߣ�HI��HBr��HCl����֮��ֻ�з��»�������Է�������Խ���»���Խ��
��3��ͼһΪƽ��ṹ�������״�ṹ��̼̼������Ϊ120����ÿ��̼ԭ�Ӷ������3��̼ԭ�ӣ�̼ԭ�Ӳ�ȡsp2�ӻ���������ÿ��Cԭ������Χ4��Cԭ���γ���������ṹ����ͼ��ʾ��
��̼ԭ�Ӱ뾶Ϊr����FA��=2r�����������ϵ��֪��AO��=4OF������FA��=3OF������AO��=![]() ����BC��Ϊa��BO��=
����BC��Ϊa��BO��=![]() ����OA��=
����OA��=![]() ���ʣ�6��1/2/3a=8/3r����a=
���ʣ�6��1/2/3a=8/3r����a=![]() �����ⳤ=
�����ⳤ=![]() �������=
�������=![]() ��������Cԭ����Ŀ=4+6��1/2+8��1/8=8������̼ԭ�������=8��4/3��r3���ʾ����ռ�������=��8��43��r3����[8/3����3��1/2r]3=34%��һ�������к�̼ԭ����Ϊ8��1/8+6��1/2+4=8����������=8��12/6.02��1023g�������е��ⳤΪ356.6pm�������=��8��12/6.02��1023g������356.6��10-10cm��3��3.5g��cm-3��
��������Cԭ����Ŀ=4+6��1/2+8��1/8=8������̼ԭ�������=8��4/3��r3���ʾ����ռ�������=��8��43��r3����[8/3����3��1/2r]3=34%��һ�������к�̼ԭ����Ϊ8��1/8+6��1/2+4=8����������=8��12/6.02��1023g�������е��ⳤΪ356.6pm�������=��8��12/6.02��1023g������356.6��10-10cm��3��3.5g��cm-3��
�ʴ�Ϊ��sp2��34%��3.5��
��4������CuΪ�����������ܶѻ������ͼ������ͭ����ľֲ��ṹ��ȷ���侧���к��м��Լ����Ǽ��Լ�����λ����
�ʴ�Ϊ�������������ܶѻ����٢ڢۡ�
��5������ͭ��Һ�м��백ˮ�������ɫ�����������μӰ�ˮ�������ܽ⣬�õ�����ɫ������Һ���йط�Ӧ�����ӷ���ʽΪCu2++2NH3��H2O�TCu��OH��2��+2NH4+��Cu��OH��2+4NH3�T[Cu��NH3��4]2++2OH-��
�ʴ�Ϊ�������γ���ɫ�����������μӰ�ˮ�������ܽ⣬�õ�����ɫ������Һ��Cu2++2NH3��H2O�TCu��OH��2��+2NH4+��Cu��OH��2+4NH3�T[Cu��NH3��4]2++2OH-��

 ѧҵ����һ��һ��ϵ�д�
ѧҵ����һ��һ��ϵ�д� Сѧ��ʱ��ҵȫͨ����ϵ�д�
Сѧ��ʱ��ҵȫͨ����ϵ�д�����Ŀ����֪��ӦH2��g��+ I2��g��![]() 2HI��g����1 mol H2��ȫ��Ӧ�ų�a kJ�������������±����ݣ�����˵������������ ��
2HI��g����1 mol H2��ȫ��Ӧ�ų�a kJ�������������±����ݣ�����˵������������ ��
�� ѧ �� | H��H | I��I |
�Ͽ�1 mol��ѧ��ʱ���յ�������kJ�� | b | c |
A���÷�Ӧ�з�Ӧ��������������������������
B���Ͽ�1 mol H-H ����1 mol I-I �������������ڶϿ�2 mol H-I ����������
C���Ͽ�2 mol H-I����������Ϊ��a+b+c��kJ
D�����ܱ������м���2 mol H2��2 mol I2����ַ�Ӧ��ų�������С��2a kJ