��Ŀ����
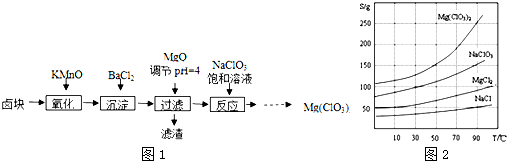
10��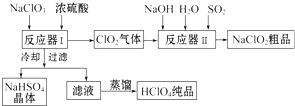 ��ҵ������������ʱ����ͬʱ����һ�ֳ�������Ҫ������������Ư���������ƣ�NaClO2���������������£�
��ҵ������������ʱ����ͬʱ����һ�ֳ�������Ҫ������������Ư���������ƣ�NaClO2���������������£���֪����NaHSO4�ܽ�����¶ȵ����߶������ʵ������¿ɽᾧ������
�ڸ�����������Ϊֹ������֪���е���ǿ�ᣬ�е�90�森��ش��������⣺
��1����ȴ��Ŀ���ǽ���NaHSO4���ܽ�ȣ�ʹNaHSO4�ᾧ������������������������ԭ���Ǹ�����ķе�ͣ���Ӧ�����з�����Ӧ�Ļ�ѧ����ʽΪ3NaClO3+3H2SO4��Ũ���THClO4+2ClO2��+3NaHSO4+H2O��
��2����Ӧ�����з�����Ӧ�����ӷ���ʽΪ2ClO2+SO2+4OH-=2ClO2-+SO42-+2H2O��
��3��ͨ�뷴Ӧ�����е�SO2����һ����H2O2���棬ͬ��������NaClO2��˫��ˮ��ʱ�����˻�ԭ�ԣ�
��4��Ca��ClO��2��ClO2��NaClO��NaClO2�Ⱥ��Ȼ����ﶼ�dz��õ���������Ư������Ϊ���Ƕ�����ǿ�����ԣ���д����ҵ����������NaOH��Һ������һ��������NaClO�����ӷ���ʽCl2+2OH-�TClO-+Cl-+H2O��
���� NaClO3��ŨH2SO4�ڷ�Ӧ��I�з�Ӧ����HClO4��ClO2��NaHSO4��ClO2�ڷ�Ӧ��II������������������Ʒ�Ӧ�����������ƣ��ٵõ��侧�壻��Ӧ��I�еõ�����Һͨ����ȴ���˵õ�NaHSO4���壬��ҺΪHClO4������õ�������HClO4��
��1���ڷ�Ӧ�����з�����Ӧ�Ŀɸ��ݼ����ԭ����NaClO3��Ũ���ᣬ��������ClO2���塢NaHSO4��HClO4��Һ��������д��ѧ����ʽ�Ĺ�����д����ѧ��Ӧ����ʽ��3NaClO3+3H2SO4=HClO4+2ClO2+3NaHSO4+H2O��ͬʱ����Ӧ��������ڵ���Һ��ȴ����NaHSO4�����������ֵõ�HClO4��Һ��˵���������Ƶ��ܽ�����¶ȵĽ��Ͷ���С��ʹ�������ƴ���Һ�нᾧ���������˺��ٽ���Һ�����ȵõ�������HClO4��˵��������ķе�ͣ�
��2������ͨ�뵽��Ӧ�������еķ�Ӧ����NaOH��SO2��H2O��ClO2����������NaClO2���ȵĻ��ϼ���+4�۱����+3�ۣ��õ����ӣ�����������ԭ��Ӧ��ԭ����Ӧʧ���ӣ���+4�۱��+6�ۣ�����������ԭ��Ӧԭ�����������õ������������ڻ�ԭ��ʧȥ���ӵ���������д��������ԭ��Ӧ����ʽ��2ClO2+SO2+4OH-=2ClO2-+SO42-+2H2O��
��3���������Ӧ�����е�SO2 ��H2O2 ����ͬ��������NaClO2��˵���任���ʺ�������ԭ��Ӧ�����ܷ�������NaClO2��Ҳ��˵��H2O2Ҳ���л�ԭ�ԣ�Ҳ�ܰ�ClO2��ԭΪNaClO2��
��4����������Ư��������ԭ����Ư��ԭ�����������ǵ�ǿ���������ﵽĿ�ģ����ݽ̲�֪ʶ��ҵ����ȡƯ��������������ʯ�ҷ�Ӧ�õ��ģ��䷴Ӧԭ��Ϊ��Cl2+2OH-=ClO-+Cl-+H2O��
��� �⣺��1������Ӧ������Ļ����Һ��ȴ�õ�NaHSO4���壬˵���������Ƶ��ܽ�����¶ȵĽ��Ͷ���С�����˺���Һ�����õ�������HClO4��˵��������ķе�ͣ�������ݼ��뵽��Ӧ�����е�ԭ����NaClO3��Ũ���ᣬ��������ClO2���塢NaHSO4��HClO4��Һ��������д��ѧ����ʽ�Ĺ���������ѧ��Ӧʱ����ԭ�ӵ��������Ŀ���䣬��д����ѧ��Ӧ����ʽΪ��3NaClO3+3H2SO4=HClO4+2ClO2+3NaHSO4+H2O��
�ʴ�Ϊ������NaHSO4���ܽ�ȣ�ʹNaHSO4�ᾧ������������ķе�ͣ�3NaClO3+3H2SO4=HClO4+2ClO2+3NaHSO4+H2O��
��2������ͨ�뵽��Ӧ�������еķ�Ӧ����NaOH��SO2��H2O��ClO2����������NaClO2���ȵĻ��ϼ���+4�۱����+3�ۣ��õ����ӣ�����������ԭ��Ӧ��ԭ����Ӧʧ���ӣ���+4�۱��+6�ۣ�����������ԭ��Ӧԭ�����������õ������������ڻ�ԭ��ʧȥ���ӵ���������������ԭ��Ӧ����ʽΪ2ClO2+SO2+4OH-=2ClO2-+SO42-+2H2O��
�ʴ�Ϊ��2ClO2+SO2+4OH-=2ClO2-+SO42-+2H2O��
��3���������Ӧ�����е�SO2 ��H2O2 ����ͬ��������NaClO2��˵���任���ʺ�������ԭ��Ӧ�����ܷ�������NaClO2��Ҳ��˵��H2O2Ҳ���л�ԭ�ԣ�Ҳ�ܰ�ClO2��ԭΪNaClO2���ʴ�Ϊ����ԭ��
��4����������Ư��������ԭ����Ư��ԭ�����������ǵ�ǿ���������ﵽĿ�ģ����ݽ̲�֪ʶ��ҵ����ȡƯ����������������������Һ��Ӧ�õ��ģ��䷴Ӧԭ��ΪCl2+2OH-=ClO-+Cl-+H2O���ʴ�Ϊ��ǿ�����ԣ�Cl2+2OH-=ClO-+Cl-+H2O��
���� ������һ���������������еĹ������̣���Ҫ����ס�������̵Ĺ��̣��ҵ���������ͻ�ƿڣ����û���֪ʶ���������Ҫ�õ��IJ�Ʒ�������Ҫ���̲���ѧ���Ļ���֪ʶӦ�õ�ʵ�ʽ��������ȥ��Ҫ�����֪ʶ�����ι̣�ͬʱ���ܽ�����֪ʶӦ��ʵ��Ӧ����

| A�� | ��������ͨ����ˮʹ����ɫ SO2+Br2+2H2O�T4H++2Br-+SO32- | |
| B�� | �Ȼ�����Һ�м�������İ�ˮ Al3++4NH3•H2O=AlO2-+4NH4++2H2O | |
| C�� | С�մ���Һ�еμӴ�����Һ HCO3-+CH3COOH=CH3COO-+H2O+CO2�� | |
| D�� | ������Һ����ˮ�����ɳ��� Al3++3H2O?Al��OH��3��+3H+ |
 ��0.8mol I2��g����1.2mol H2��g������ij1L�ܱ������У���һ���¶��·�����Ӧ��I2��g��+H2��g��?2HI��g�����ﵽƽ�⣮HI�����������ʱ��ı仯�������ʾ��
��0.8mol I2��g����1.2mol H2��g������ij1L�ܱ������У���һ���¶��·�����Ӧ��I2��g��+H2��g��?2HI��g�����ﵽƽ�⣮HI�����������ʱ��ı仯�������ʾ��| HI������� | 1min | 2min | 3min | 4min | 5min | 6min | 7min |
| ����I | 26% | 42% | 52% | 57% | 60% | 60% | 60% |
| ����II | 20% | 33% | 43% | 52% | 57% | 65% | 65% |
��2��������I�ӿ�ʼ��Ӧ������ƽ��ʱ��H2�ķ�Ӧ����Ϊ0.12 mol/��L•min����
��3��Ϊ�ﵽ����II�����ݣ����ڷ�Ӧ��ϵ���ܸı�IJ����ǽ����¶ȣ�
��4���÷�Ӧ�ġ�H��0�����������������=����
��5��������I�´ﵽƽ�����7minʱ���������ѹ��Ϊԭ����һ�룮����ͼ�л���c��HI����ʱ��仯�����ߣ�
| A�� | Fe��OH��3��������Fe��OH��3+3H+�TFe3++3H2O | |
| B�� | ����������Һ��ͨ������������壺2ClO-+SO2+H2O�T2HClO+SO32- | |
| C�� | ���������Һ��ͨ������Ķ�����̼��Ca2++2ClO-+H2O+CO2�TCaCO3��+2HClO | |
| D�� | ����NaHSO4�����Ba��OH��2��Һ��Ӧ��H++SO42-+Ba2++OH-�TBaSO4��+H2O |
| A�� | 1 mol����������Ϊ32 g | |
| B�� | ���³�ѹ�£�1 mol CO2�������ԼΪ22.4 L | |
| C�� | CO2��Ħ������Ϊ44 g•mol-1 | |
| D�� | 1 L 2 mol•L-1��BaCl2��Һ�к�Cl-�ĸ���Ϊ2.408��1024 |