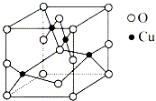
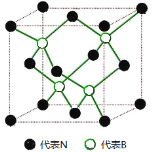
��Ŀ����
����Ŀ��I��ʵ������Ҫ0.1mol/L������Һ490mL���ֽ������ƣ����ں�������д�ʵ����������ƣ��������������֡�
��1������Ͳ��ȡ�ܶ�Ϊ1.19g/cm3��������Ϊ36.5%��Ũ����_______mLע���ձ��С�
��2����ʢ������ձ���ע����������ˮ���ò��������裬ʹ����ȡ�
��3����ϡ�͵�������ȴ���ز�����ע��______�С�
��4��������ˮϴ���ձ���������2��3�Σ���ϴ��Һע������ƿ�С�
��5��������ƿ��С�ļ�����ˮ��Һ��ӽ��̶���______cm��������______�μ�����ˮ��ʹ��Һ��Һ�����ʹ�ǡ����̶������С�
����ʵ�������������������������������Һ�����ʵ���Ũ���к�Ӱ�죨��ƫ�ߡ�ƫ�͡���Ӱ�죩��
��1��δ��ϴ��Һ��������ƿ______
��2������ǰ����ƿ��ˮϴ�Ӻ�δ���и��ﴦ��_______��
��3������������Һδ��ȴ�����¾�ת�Ƶ�����ƿ_________��
���𰸡� 4.2mL 500 mL����ƿ 1��2 ��ͷ�ι� ƫ�� ��Ӱ�� ƫ��
��������I����1��ʵ����û��490mL����ƿ�������Ҫ����500mL���ܶ�Ϊ1.19g/cm3��������Ϊ36.5%��Ũ��������ʵ���Ũ����![]() ��������Ͳ��ȡŨ��������Ϊ
��������Ͳ��ȡŨ��������Ϊ![]() ����3����ϡ�͵�������ȴ���ز�����ע��500mL����ƿ�С���5��������ƿ��С�ļ�����ˮ��Һ��ӽ��̶���1��2cm�������ý�ͷ�ιܵμ�����ˮ��ʹ��Һ��Һ�����ʹ�ǡ����̶������С�
����3����ϡ�͵�������ȴ���ز�����ע��500mL����ƿ�С���5��������ƿ��С�ļ�����ˮ��Һ��ӽ��̶���1��2cm�������ý�ͷ�ιܵμ�����ˮ��ʹ��Һ��Һ�����ʹ�ǡ����̶������С�
��1��δ��ϴ��Һ��������ƿ�У����ʼ��٣�Ũ��ƫ�ͣ���2������ǰ����ƿ��ˮϴ�Ӻ�δ���и��ﴦ������Ӱ�죻��3������������Һδ��ȴ�����¾�ת�Ƶ�����ƿ�У���������������֪��Һ������٣�Ũ��ƫ�ߡ�