��Ŀ����
����������Ӧ���Ȼ�ѧ����ʽ��
��I2(g) + H2(g) ��2HI(g)��������H =��9.48 kJ��mol��1��
��2HI(g)��������H =��9.48 kJ��mol��1��
��I2(s) + H2(g) ��2HI(g)���� �� ��H =" +26.48" kJ��mol��1
��2HI(g)���� �� ��H =" +26.48" kJ��mol��1
����˵����ȷ���ǣ� ��
��I2(g) + H2(g)
 ��2HI(g)��������H =��9.48 kJ��mol��1��
��2HI(g)��������H =��9.48 kJ��mol��1�� ��I2(s) + H2(g)
 ��2HI(g)���� �� ��H =" +26.48" kJ��mol��1
��2HI(g)���� �� ��H =" +26.48" kJ��mol��1����˵����ȷ���ǣ� ��
| A���ٵIJ���ȢڵIJ����ȶ� |
| B��I2(s) = I2(g)��������H=" +17.00" kJ��mol��1 |
| C���ڵķ�Ӧ���������Ȣٵķ�Ӧ���������� |
| D��1mol I2(g)��ͨ��1 mol H2(g)��������Ӧʱ����9.48 kJ |
C
����������ٵIJ���͢ڵIJ��ﶼ��HI���ȶ�����ͬ��A����ȷ���ؼ���˹���ɿ�֪���ڣ��ټ��õ�I2(s)��I2(g)����H��26.48kJ/mol��9.48kJ/mol����35.96kJ/mol��ѡ��B����ȷ������������������̬�ĵ����������Ԣڵķ�Ӧ���������Ȣٵķ�Ӧ���������ͣ�C��ȷ���÷�Ӧ�ǿ��淴Ӧ����1mol I2(g)��ͨ��1 mol H2(g)��������Ӧʱ����һ��С��9.48kJ��D����ȷ����ѡC��
�����������Ǹ߿��еij������ͣ������е��Ѷȵ����⡣��Ҫ�ǿ���ѧ���Է�Ӧ�Ⱥ����Լ����ø�˹���ɼ��㷴Ӧ�ȵ��˽����������ּ�ڹ���ѧ���Ļ��������ѧ����Ӧ��������ѧϰЧ�ʡ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
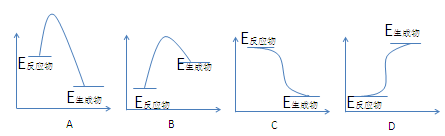
 CH3OH(g)�����е������仯�������ͼ��ʾ�����ߢ�����ߢ�ֱ��ʾ��ʹ�ô�����ʹ�ô�������������������ж���ȷ����
CH3OH(g)�����е������仯�������ͼ��ʾ�����ߢ�����ߢ�ֱ��ʾ��ʹ�ô�����ʹ�ô�������������������ж���ȷ����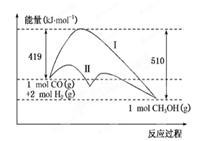
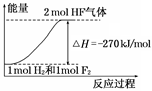
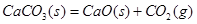 �����²����Է����У�˵���÷�Ӧ��
�����²����Է����У�˵���÷�Ӧ��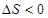
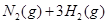


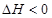 ��������������ʱ�����¶ȣ���Ӧ����v��H2����v��NH3��������
��������������ʱ�����¶ȣ���Ӧ����v��H2����v��NH3�������� C(g) +D(g)��H= ��kj.mol-1�ķ�Ӧ����(����꣩���������ݱ�/kj���ı仯��ϵ��ͼ��ʾ��������ȷ����
C(g) +D(g)��H= ��kj.mol-1�ķ�Ӧ����(����꣩���������ݱ�/kj���ı仯��ϵ��ͼ��ʾ��������ȷ����
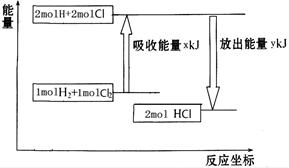
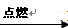 2HCl + Q
2HCl + Q