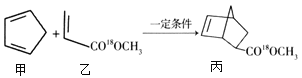��Ŀ����
����Ŀ�����¶ȡ��ݻ���ͬ��3���ܱ������У�����ͬ��ʽͶ�뷴Ӧ����ֺ��¡����ݣ���÷�Ӧ�ﵽƽ��ʱ���й���������[��֪N2(g)��3H2(g)![]() 2NH3(g)����H����92.4 kJ��mol��1]������˵����ȷ����(����)
2NH3(g)����H����92.4 kJ��mol��1]������˵����ȷ����(����)
���� | �� | �� | �� |
��Ӧ��Ͷ���� | 1 mol N2�� 3 mol H2 | 2 mol NH3 | 4 mol NH3 |
NH3��Ũ ��/mol��L��1 | c1 | c2 | c3 |
��Ӧ�������仯 | �ų�a kJ | ����b kJ | ����c kJ |
��ϵѹǿ(Pa) | p1 | p2 | p3 |
��Ӧ��ת���� | ��1 | ��2 | ��3 |
A. 2c1��c3B. ��1����3��1C. 2p2��p3D. a��b��92.4
���𰸡�B
��������
A���ס�����Ƚϣ��Ѽ�ЧΪ��ʼ����2 mol NH3�����а��������ʵ���Ϊ��2��������������Ϊ�������Ϊ��2���������NH3�����ʵ���Ϊ��2����Ȼ���С������������ͬ��ѹǿ�����ڷ�ӦN2(g)��3H2(g)![]() 2NH3(g)����ƽ�������ɰ����ķ����ƶ�����2c1��c3����A����
2NH3(g)����ƽ�������ɰ����ķ����ƶ�����2c1��c3����A����
B���ס��Ҵ�����ͬ��ƽ��״̬�����1+��2=1���ҡ�������൱�ڱ�����ѹǿ��N2(g)��3H2(g)![]() 2NH3(g)ƽ�������ƶ�������ת���ʼ�С����2����3��������1����3��1����B��ȷ��
2NH3(g)ƽ�������ƶ�������ת���ʼ�С����2����3��������1����3��1����B��ȷ��
C���Ƚ��ҡ�����֪�����а��������ʵ���Ϊ�ҵ�2��������������Ϊ�������Ϊ��2���������NH3�����ʵ���Ϊ��2����Ȼ���С�������������ͬ����ƽ�ⲻ�ƶ���2p2=p3��ѹǿ�����ڷ�ӦN2(g)��3H2(g)![]() 2NH3(g)����ƽ�������ɰ����ķ����ƶ�������2p2��p3����C���� ��
2NH3(g)����ƽ�������ɰ����ķ����ƶ�������2p2��p3����C���� ��
D���ס���ʵ����Ϊ��Чƽ�⣬��ͬ���Ƿ�Ӧ����ʼ����ͬ���ڴ˹����������յ������൱�ڼ���ȫת�����ٷų�����������a+b=92.4����D����ѡB��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ����20 L���ܱ������а����ʵ���֮��Ϊ1��2����CO��H2��������Ӧ��CO(g)��2H2(g)![]() CH3OH(g)����H�����CO��ת�������¶ȼ���ͬѹǿ�µı仯��ͼ��ʾ��p2��195 ��ʱn(H2)��ʱ��ı仯��������ʾ������˵����ȷ����(����)
CH3OH(g)����H�����CO��ת�������¶ȼ���ͬѹǿ�µı仯��ͼ��ʾ��p2��195 ��ʱn(H2)��ʱ��ı仯��������ʾ������˵����ȷ����(����)
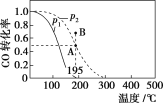
p2��195 �� ʱn(H2)��ʱ��仯
t/min | 0 | 1 | 3 | 5 |
n(H2)/mol | 8 | 5 | 4 | 4 |
A. p1��p2����H��0
B. ��p2��195 �� ʱ����Ӧǰ3 min��ƽ������v(CH3OH)��0.8 mol��L��1��min��1
C. ��p2��195 �� ʱ���÷�Ӧ��ƽ�ⳣ��Ϊ25
D. ��B ��ʱ��v����v��
����Ŀ��ʵ������63%��Ũ���ᣨ���ܶ�Ϊ1.4g/mL������240mL0.50mol/Lϡ���ᣬ��ʵ�������У�A 10mL��Ͳ B 50mL��Ͳ C ������ƽ D ������ E 100mL����ƿ F 250mL����ƿ G 500mL����ƿ H ��ͷ�ι� I 200mL�ձ���
��1����Ũ��������ʵ���Ũ��Ϊ____________mol/L
��2��Ӧ��ȡ63%��Ũ����_____________mL��Ӧѡ��___________��(����������ĸ���)
��3��ʵ��ʱ����ѡ�õ�������______________������������ĸ��ţ�
��4�����ƹ����У����в�����ʹ���Ƶ�ϡ������ҺŨ��ƫ�ߵ���______������ţ��� |
A�������˽���Ľ�������ʹ������ƿ |
B�����ܰ��˽���Ľ�����Ϊ�ή������ƿ�ľ�ȷ�� |
C�����ؼӴ�ƿ������Ϊ������ƿ��ת��Һ��ʱ��������Һ�嵹��ƿ�⣬�������Һ��Ũ�Ȳ���̫��Ӱ�� |