��Ŀ����
��ѧ��Ӧԭ���ڿ��к��������й㷺Ӧ��
��1�����á���ѧ����ת�Ʒ����Ʊ�TaS2���壬�������·�ӦTaS2(s)+2I2(g) TaI4(g)+S2(g) ��H��0 ����Ӧ����ƽ�ⳣ������ʽK= ����K=1����ij�����ܱ������м���1mol I2��g��������TaS2��s����I2��g����ƽ��ת����Ϊ ��
TaI4(g)+S2(g) ��H��0 ����Ӧ����ƽ�ⳣ������ʽK= ����K=1����ij�����ܱ������м���1mol I2��g��������TaS2��s����I2��g����ƽ��ת����Ϊ ��
��2����ͼ��ʾ����Ӧ������ʯӢ��չ��н��У������¶�ΪT2��һ�˷���δ�ᴿ��TaS2��ĩ������I2��g����һ��ʱ������¶�ΪT1��һ�˵õ��˴�����TaS2���壬���¶�T1 T2���������������=������������Ӧ��ϵ��ѭ��ʹ�õ������� ��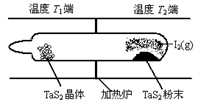
��3������I2�������Կɲⶨ��������ĺ����������ǽ������е���ת��Ϊ H2SO3��Ȼ����һ��Ũ�ȵ�I2��Һ���еζ�������ָʾ��Ϊ ���ζ���Ӧ�����ӷ���ʽΪ ��
��4��25��ʱ��H2SO3 HSO3-+H+�ĵ��볣��Ka=1��10-2mol/L������¶���NaHSO3��ˮ��ƽ�ⳣ��Kh= mol/L������NaHSO3��Һ�м���������I2������Һ��
HSO3-+H+�ĵ��볣��Ka=1��10-2mol/L������¶���NaHSO3��ˮ��ƽ�ⳣ��Kh= mol/L������NaHSO3��Һ�м���������I2������Һ�� �� ���������С�����䡱����
�� ���������С�����䡱����
��1��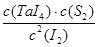 ��66.7%��
��66.7%��
��2������ I2��
��3�����ۣ� H2SO3+I2+H2O=2I��+SO42��+4H����
��4��1.0��10��12������
����

��ѧ��Ӧԭ���ڿ��к��������й㷺Ӧ�á�
����ͼװ����ʾ��A��B�еĵ缫Ϊ��Ķ��Ե缫��C��DΪ���ڽ���Na2SO4��Һ����ֽ���ϵIJ��У���Դ��a��b��������A��B�г���KOH��Һ������KOH��Һ��ˮ���С��ж�K1���պ�K2��K3ֱͨ���硣

�ش��������⣺
��1�� a�ǵ�Դ�� ����д��A�еĵ缫��ӦʽΪ_____��
��2��ʪ��Na2SO4��ֽ�����ܹ۲쵽��������____________��
��3�����һ��ʱ���A��B�о��������Χ�缫������ʱ�ж�K2��K3���պ�K1�����ֵ�������ָ���ƶ���д����ʱB�еĵ缫��ӦʽΪ ��
�״���һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ������ҵ��һ��������з�Ӧ�ϳɼ״���CO(g)��2H2(g)
 CH3OH(g)
��H���±����������Ǹ÷�Ӧ�ڲ�ͬ�¶��µĻ�ѧƽ�ⳣ��(K)����ش��������⣺
CH3OH(g)
��H���±����������Ǹ÷�Ӧ�ڲ�ͬ�¶��µĻ�ѧƽ�ⳣ��(K)����ش��������⣺
|
�¶� |
250�� |
300�� |
350�� |
|
K |
2.041 |
0.270 |
0.012 |
��4���ɱ��������жϦ�H 0�����������������������
��5�������������䣬ֻ�ı�����һ�����������д�ʩ����״����ʵ��� ��
A�������¶ȣ�B��ʹ�ú��ʵĴ�����C����С�������ݻ���D�����������H2��E����ѹʱ������He��F������ϵ�з����CH3OH
��6��ij�¶��£���2mol CO��6 mol H2����2L�ܱ������У���Ӧ���е�4minĩ�ﵽƽ�⣬��ʱ���c(CO) ��0.2 mol/L ����0~4min��H2�ķ�Ӧ����Ϊ ���������¶��ݻ������������г���һ������CH3OH�����´ﵽ��ѧƽ��״̬����ԭƽ��״̬��ȣ���ʱƽ����������CH3OH��������� ����������С�������䡱����
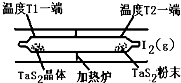
 ��2����ͼ��ʾ����Ӧ��I����ʯӢ��չ��н��У������¶�ΪT2��һ�˷���δ�ᴿ��TaS2��ĩ������I2��g����һ��ʱ������¶�ΪT1��һ�˵õ��˴�����TaS2���壬���¶�T1 T2���������������=������������Ӧ��ϵ��ѭ��ʹ�õ������� ��
��2����ͼ��ʾ����Ӧ��I����ʯӢ��չ��н��У������¶�ΪT2��һ�˷���δ�ᴿ��TaS2��ĩ������I2��g����һ��ʱ������¶�ΪT1��һ�˵õ��˴�����TaS2���壬���¶�T1 T2���������������=������������Ӧ��ϵ��ѭ��ʹ�õ������� �� TaI4��g��+S2��g�� ����
TaI4��g��+S2��g�� ����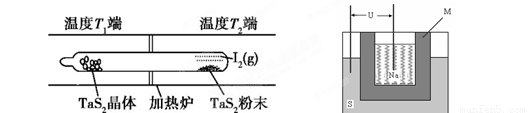
 TaI4(g)+S2(g) ��H��0 ����Ӧ����ƽ�ⳣ������ʽK= ����K=1����ij�����ܱ������м���1mol I2��g��������TaS2��s����I2��g����ƽ��ת����Ϊ ��
TaI4(g)+S2(g) ��H��0 ����Ӧ����ƽ�ⳣ������ʽK= ����K=1����ij�����ܱ������м���1mol I2��g��������TaS2��s����I2��g����ƽ��ת����Ϊ ��
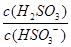 �� ���������С�����䡱����
�� ���������С�����䡱����