��Ŀ����
����ΪһЩ������Ԫ�ؼ���Ӧ�⻯��е�����ݣ�
Ԫ�� �� �ԡ� �� | ������������ Ԫ �� �� �š����������� | |||||
�� | �� | �� | �� | �� | �� | |
�⻯��ķе㣨�棩 | -60.7 | -33.4 | 100 | -87.7 | 19.54 | -84.9 |
�� �� �� �� �� | +6 | +5 | +5 | +7 | ||
�� �� �� �� �� | -2 | -3 | -2 | -3 | -1 | -1 |
������A��B��Y��Ϊ�ϱ��е�Ԫ�ء�X��һ����ʷ�ƾã�Ӧ�ù㷺�Ľ���Ԫ�ء�
�� X��Y���γɻ�����XY��XY2, ���߿��ýӴ�����ǿ��ף�
�� A��B���γɻ�����AB��AB2�����߾��������Ʊ�ǿ���ҡ�
��ش�
��1��Ԫ�� �� �⻯��ķ���ʽ�� __________��
��2�������й�ǿ��ס���˵����ȷ���� ________��
a�� ���ߵ�Ũ��Һ�ڳ���ʱ���������ƻ����Ƶ���������
b�� ���ߵ�Ũ��Һ�ڳ��������з��ã���������仯
c�� ��ҵ����ǿ��ס���ʱ����Ҫ��ˮ������Ӧ��������
d�� ���ߵ�ϡ��Һ����ǿ������
��3����ҵ����ǿ���ʱ����Ũ��ˮ����β���������ij����Ρ�
��ѧ����ʽΪ__________________________________��
��4��д����ҵ����ǿ����ʱ���AB�Ļ�ѧ����ʽ������������������������������ ��
��5����ҵ����ǿ����ʱ���ô�����Һ����β�����÷�Ӧ���£�
AB + AB2 + Na2CO3 = 2 ��![]() ��+ CO2
��+ CO2
�� �ڷ�������ij�ε���ѧʽ
�� ÿ����44.8 L����״����CO2��������β���������� ______�硣
��6�� �������ڵ������ڵ�Ԫ����_______________���ñ���Ԫ�ر����д����
��1�� H2O��������������������������������������������������
��2�� a b������
��3�� 2NH3?H2O + SO2 = (NH4)2SO3 + H2O��������������������
��4�� 4NH����5O2������ 4NO + 6H2O����������������
��5����NaNO2������������ �� 15
��6���١��ܡ��ޡ���

����ΪһЩ������Ԫ�ؼ���Ӧ�⻯��е�����ݣ�
| ||||||||||||||||||||||||||||||||||
��11�֣�
��1�����±���һЩ������Ԫ�ص���̬ԭ��ʧȥ���ⲻͬ�������������(kJ��mol-1)��
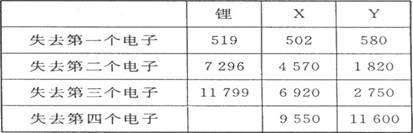
ͨ�����е����ݷ���Ϊʲô�ԭ��ʧȥ����ڶ�������ʱ���������ҪԶԶ����ʧȥ��һ��������������� ��
X�����ڱ���λ�ã��� �ڣ� �壬Y���������Ϊ ��
��2��1932��������ѧ�ұ��֣�L��Pauling����������˵縺�Եĸ���縺�ԣ���X��ʾ��Ҳ��Ԫ�ص�һ����Ҫ���ʣ��±���������ԭ������С��20��16��Ԫ�صĵ縺����ֵ��
|
Ԫ�� |
H |
Li |
Be |
B |
C |
N |
O |
F |
|
�縺�� |
2.1 |
1.0 |
1.5] |
2.0 |
2.5 |
3.0 |
3.5 |
4.0 |
|
Ԫ�� |
Na |
Mg |
Al |
Si |
P |
S |
Cl |
K |
|
�縺�� |
0.9 |
1.2 |
1.5 |
1.7 |
2.1 |
2.3 |
3.0 |
0.8 |
����ϸ�������ش������й����⣺
�١��ϱ��е縺����С��Ԫ���� (��Ԫ�ط���)�����Ƹ�Ԫ�صĵ縺�Ե�ȡֵ��Χ��__________��X��__________��
�ڡ�������ɸ������ǣ����γɻ�ѧ������ԭ����ӦԪ�صĵ縺�Բ�ֵ����1.7ʱ�����γ� ��һ��Ϊ���Ӽ�����С��1.7ʱ��һ��Ϊ���ۼ������ƶ�AlBr3���γɵĻ�ѧ��������Ϊ
______________���������� ��
| Ԫ������ | Ԫ�ر�� | |||||
| �� | �� | �� | �� | �� | �� | |
| �⻯��ķе㣨�棩 | -60.7 | -33.4 | 100 | -87.7 | 19.54 | -84.9 |
| ����ϼ� | +6 | +5 | +5 | +7 | ||
| ��ͻ��ϼ� | -2 | -3 | -2 | -3 | -1 | -1 |
A�����ߵ�Ũ��Һ�ڳ���ʱ������X�ĵ����Ƴ���������
B����ҵ����ǿ���ҵĵ�һ���ǽ��ڵ��⻯���ڴ�����ȼ����������AB
C����ҵ����ǿ��ס���ʱ����Ҫ��ˮ������Ӧ��������
D���٢ڢ۵��⻯����ܱ�ǿ���Ũ��Һ����