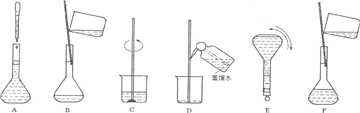
��Ŀ����
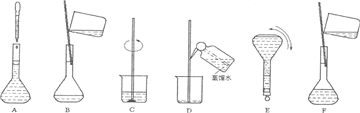
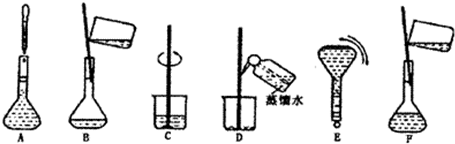
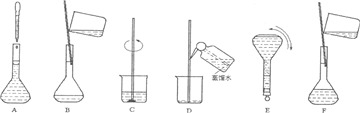
��ͼ�������Լ�ƿ��ǩ�ϵ����ݣ�

(1)����������ʵ���Ũ����________mol/L��
(2)ij��ѧ��ȤС������������ʵ�ʵ��̽��ʱ����Ҫ240 mL��4.6 mol/L��ϡH2SO4������Ҫȡ________mL�ĸ����ᣬ�������������ϡ�͵�ʵ�����Ϊ________��
(3)������4.6 mol/LϡH2SO4�Ĺ����У����������������Һ���ʵ���Ũ���к�Ӱ��(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)��
��δ����ȴ������Һע������ƿ�У�________��
������ƿ��1 mol/LϡH2SO4��ϴ��________��
�۶���ʱ���ӹ۲�Һ��________��
(4)�����£�ijѧ��ȡ�������Լ�ƿ�е�������һ�ྻ�Թ��У����뼸Ƭ���������Ƭ����û�����ݲ�����Ҳδ������Ƭ�ܽ⣬������ͬѧ������ԭ��________��
�𰸣�

��ϰ��ϵ�д�
�����Ŀ