��Ŀ����
��7�֣�(1)��֪��H��H����Ϊ436kJ/mol��N��N����Ϊ945kJ/mol��N��H����Ϊ391kJ/mol�����ݼ��ܼ��㹤ҵ�ϳɰ��ķ�Ӧ��___________ ��Ӧ(����ȡ����ȡ�)������1mol N2�ϳɰ���Ӧ��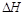 =______________��
=______________��
(2) �ָ�������3���Ȼ�ѧ��Ӧ����ʽ��
��Fe2O3(s)+3CO(g) ��2Fe(s)+3CO2(g) ��H���D24.8kJ��mol
��3Fe2O3(s)+ CO(g) ��2Fe3O4(s)+ CO2(g) ��H���D47.2kJ��mol
��Fe3O4(s)+CO(g) ��3FeO(s)+CO2(g) ��H��+640.5kJ��mol
��CO���廹ԭFeO����õ�Fe�����CO2������Ȼ�ѧ��Ӧ����ʽΪ ��
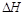 =______________��
=______________�� (2) �ָ�������3���Ȼ�ѧ��Ӧ����ʽ��
��Fe2O3(s)+3CO(g) ��2Fe(s)+3CO2(g) ��H���D24.8kJ��mol
��3Fe2O3(s)+ CO(g) ��2Fe3O4(s)+ CO2(g) ��H���D47.2kJ��mol
��Fe3O4(s)+CO(g) ��3FeO(s)+CO2(g) ��H��+640.5kJ��mol
��CO���廹ԭFeO����õ�Fe�����CO2������Ȼ�ѧ��Ӧ����ʽΪ ��
��1�� ����(2��)����93KJ/mol (2��)
��2��CO (g)��FeO(s)=" Fe(s)+" CO2 (g) ��H=��218KJ/mol (3��)
��2��CO (g)��FeO(s)=" Fe(s)+" CO2 (g) ��H=��218KJ/mol (3��)
�����������1����Ӧ�ȵ��ڷ�Ӧ����ܼ��ܼ�ȥ��������ܼ���945kJ/mol+3��436KJ/mol-6��391kJ/mol=��93KJ/mol ��ͨ�����㷴Ӧ�ġ�H����0Ϊ���ȷ�Ӧ����2�������Ȼ�ѧ����ʽΪ-��-��-2���ۣ���CO (g)��FeO(s)=" Fe(s)+" CO2 (g)����H����Ӧ�ļ��㣬��H=��218KJ/mol��
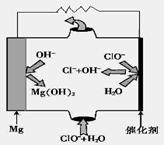
��������������ע���˹���ɵ�Ӧ�á����ڼ��⡣

��ϰ��ϵ�д�
 ��ѧ��ʦ����ϵ�д�
��ѧ��ʦ����ϵ�д�
�����Ŀ
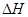 �ľ���ֵ����ȷ��
�ľ���ֵ����ȷ��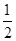 O2(g)===H2O(l)����H3����285.8 kJ/mol
O2(g)===H2O(l)����H3����285.8 kJ/mol
 ��
�� 

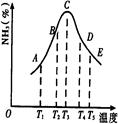
 2NH3�����ֱ���t��ʱ�ⶨ����NH3�������������ͼ���ң�
2NH3�����ֱ���t��ʱ�ⶨ����NH3�������������ͼ���ң�