��Ŀ����
��14�֣����ܱ������н������·�Ӧ��3m A��g��+m B��g�� n C��g��+q D��s����
n C��g��+q D��s����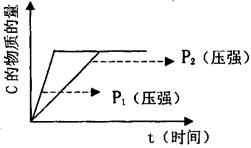
��һ��
��1��������ʼ�ݻ���ȵ���������ɱ�������У��������ʵ�����ȵķ�Ӧ���Ӧ�����б�����ϵ���¶Ȳ��䣩��������ͼ��ʾ��ƽ�⣬����ͼ��֪��P1 P2���>������<����=������m��n֮��Ĺ�ϵΪ�� ���ú�m�Ĵ���ʽ��ʾn����
��2���ԣ�1���ķ�Ӧ���ԣ������ܹ��жϷ�Ӧ�Ѵﻯѧƽ��״̬���� ��
A�������ܵ����ʵ������� B������D�����ʵ���һ��
C��������ܶȲ��� D��������A��B�����ʵ���֮��Ϊ3:1ʱ
E������A��Ũ�Ȳ��� F����Ӧ����v ��A��=3m/nv��C��
��3����һ�����¶Ⱥ�ѹǿ�£���3moIA��1moIB��������ɱ���������ﵽƽ��ʱ��B�����ʵ���Ũ��Ϊamol/L���ֳ�������ѹǿ���У�
�ٵ�ѹǿΪƽ��ʱ��2��5��ʱ��Bƽ��ʱ�����ʵ���Ũ��ΪC1 mol/L,���C1=-2.5a��
�ڵ�ѹǿΪƽ��ʱ��10��ʱ�� Bƽ��ʱ�����ʵ���Ũ��ΪC2mol/L�����C2= 5.5a;
�Է�����ɵڢ��ֽ���Ŀ���ԭ�� ��
��������m=n=q���ұ���ԭ������������䣬��һ���¶��£���3molA��1mnolB����������У�ƽ��ʱ������B��ת����ΪW%��
��4������������¶Ȳ���������£����������↑ʼ���ϣ�Ҫ�ﵽ������������D����ʼ���ʵ���n��D��Ӧ�����������____ ���ú���W%�Ĵ���ʽ��ʾ����ͬ����ƽ��ʱC��ת����Ϊ ��
��1��P1>P2 n="4m " ����2�֣�
��2��BCE ��4�� ��
��
��3��C��Ϊ����̬���� ��2�֣�
��4��n��D��>1 һ w�� 1һW�� ����2�֣�
����

| A��ZΪ0.3 mol/L | B��YΪ0.15 mol/L | C��XΪ0.1 mol/L | D��ZΪ0.4 mol/L |
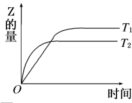
| A��T1��T2������Ӧ�Ƿ��ȷ�Ӧ | B��T1��T2������Ӧ�����ȷ�Ӧ | C��T1��T2������Ӧ�Ƿ��ȷ�Ӧ | D��T1��T2������Ӧ�����ȷ�Ӧ |
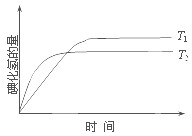 ���ܱ������н������·�Ӧ��H2��g��+I2��g��?2HI��g������H�����¶�T1��T2ʱ����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ������ͼʾ����ȷ�ж��ǣ�������
���ܱ������н������·�Ӧ��H2��g��+I2��g��?2HI��g������H�����¶�T1��T2ʱ����������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ������ͼʾ����ȷ�ж��ǣ�������