题目内容
已知热化学方程式:
4Al(s)+3O2(g)═2Al2O3 (s)△H1=-3288.6kJ?mol-1,
4Fe(s)+3O2(g)═2Fe2O3 (s)△H2=-1631.8kJ?mol-1,
则铝粉与氧化铁发生铝热反应的热化学方程式为
4Al(s)+3O2(g)═2Al2O3 (s)△H1=-3288.6kJ?mol-1,
4Fe(s)+3O2(g)═2Fe2O3 (s)△H2=-1631.8kJ?mol-1,
则铝粉与氧化铁发生铝热反应的热化学方程式为
2Al(s)+Fe2O3 (s)═Al2O3 (s)+2Fe(s)△H=-828.4KJ/mol
2Al(s)+Fe2O3 (s)═Al2O3 (s)+2Fe(s)△H=-828.4KJ/mol
.分析:依据热化学方程式和盖斯定律计算得到所需热化学方程式,注意标注物质的聚集状态和对应反应的焓变.
解答:解:①4Al(s)+3O2(g)═2Al2O3 (s)△H1=-3288.6kJ?mol-1;
②4Fe(s)+3O2(g)═2Fe2O3 (s)△H2=-1631.8kJ?mol-1;
由盖斯定律计算①-②得到:4Al(s)+2Fe2O3 (s)═2Al2O3 (s)+4Fe(s)△H=-1656.8KJ/mol
热化学方程式为:2Al(s)+Fe2O3 (s)═Al2O3 (s)+2Fe(s)△H=-828.4KJ/mol
故答案为:2Al(s)+Fe2O3 (s)═Al2O3 (s)+2Fe(s)△H=-828.4KJ/mol.
②4Fe(s)+3O2(g)═2Fe2O3 (s)△H2=-1631.8kJ?mol-1;
由盖斯定律计算①-②得到:4Al(s)+2Fe2O3 (s)═2Al2O3 (s)+4Fe(s)△H=-1656.8KJ/mol
热化学方程式为:2Al(s)+Fe2O3 (s)═Al2O3 (s)+2Fe(s)△H=-828.4KJ/mol
故答案为:2Al(s)+Fe2O3 (s)═Al2O3 (s)+2Fe(s)△H=-828.4KJ/mol.
点评:本题考查了热化学方程式的书写方法和盖斯定律的计算应用,题目较简单.

练习册系列答案
相关题目
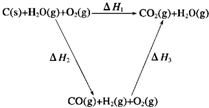
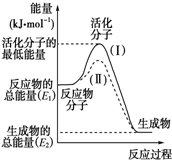 (1)图中所示反应是
(1)图中所示反应是