��Ŀ����
����Ŀ��ij��ѧС���Ա����ᣨ ��Ϊԭ�ϣ���ȡ�������������֪�й����ʵķе������
��Ϊԭ�ϣ���ȡ�������������֪�й����ʵķе������
���� | �״� | ������ | ��������� |
�е�/�� | 64.7 | 249 | 199.6 |
�ܶ�/g��cm-3 | 0.792 | 1.2659 | 1.0888 |
��Է������� | 32 | 122 | 136 |
��.�ϳɱ���������ֲ�Ʒ
��Բ����ƿ�м���12.2g�������20mL�״�(�ܶ�ԼΪ0.79g��cm��3)����С�ļ���3mLŨ���ᣬ���Ⱥ�Ͷ�뼸�����Ƭ��С�ļ���ʹ��Ӧ��ȫ���ñ���������ֲ�Ʒ��
��1��Ũ�����������______________��д����������״���Ӧ�Ļ�ѧ����ʽ��______________��
��2��������λͬѧ�ֱ��������ͼ��ʾ������ʵ���Һϳɱ����������װ��(�г������ͼ�������������ȥ)�������л���ķе㣬��ò���__________װ��(����������������)

��3����Ӧ��CH3OHӦ������������______________��
��.�ֲ�Ʒ�ľ���
��4������������ֲ�Ʒ���������������״����������ˮ�ȣ���������������ͼ���о��ƣ���������ͼ�з������������������������a______________��b______________��
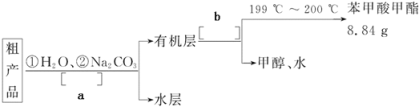
��5��ͨ�����㣬����������IJ���Ϊ______________
���𰸡���������ˮ�� ![]() +CH3OH
+CH3OH![]()
![]() +H2O �� ��������߱������ת���� ��Һ ���� 65%
+H2O �� ��������߱������ת���� ��Һ ���� 65%
��������
������ͼ״���Ũ���ᡢ���������·���������Ӧ���÷�Ӧ�ǿ��淴Ӧ�������ִ��ڸ���Ӧ���ʵñ���������ֲ�Ʒ������������ֲ�Ʒ���������������״����������ˮ�ȣ��ʿ��뱥��̼������Һ��ϣ���������Ϊ�����ͱ�����������ܽ�ȣ��ܽ�״�����ȥ�ᣬ����Һ��õ��ϴ����ı������������һ�������ô���Ʒ��
��. (1)Ũ������������Ӧ�����˴������ú���ˮ�������ã���������״���Ӧ�Ļ�ѧ����ʽ��![]() +CH3OH
+CH3OH![]()
![]() +H2O ��
+H2O ��
��Ϊ����������ˮ����![]() +CH3OH
+CH3OH![]()
![]() +H2O��
+H2O��
(2)��װ��ͼ��֪����ͼ��Բ����ƿ���������ܣ�����������ã���ͼ��û�У��������з�Ӧ��״��е�ͣ������ᡢ����������ķе�Զ���ڼ״�����������ͼ���״��ض�������ӷ��������ںϳɷ�Ӧ������Ӧ�������������ټ״��Ļӷ�����߲��ʣ�
��Ϊ���ף�
(3)������ӦΪ���淴Ӧ������Ӧ���Ũ�ȣ�ƽ��������Ӧ�����ƶ����ʼ״�Ӧ��������Ϊ����߱������ת���ʣ�
��.(4)���������������ˮ�������������������ܵ�Һ�壬ͨ�����÷�Һ������ɣ��״��ͱ����������ܽ⣬���߷е㲻ͬ������ͨ������������룻
��Ϊ����Һ������
(5)12.2g����������ʵ���Ϊ��![]() ��20mL�״�(�ܶ�Լ0.79g/mL)�����ʵ���Ϊ��
��20mL�״�(�ܶ�Լ0.79g/mL)�����ʵ���Ϊ��![]() �������������ɱ�������������ʵ���Ϊ0.1mol������Ϊ��136g/mol��0.1mol=13.6g����������IJ���Ϊ��
�������������ɱ�������������ʵ���Ϊ0.1mol������Ϊ��136g/mol��0.1mol=13.6g����������IJ���Ϊ�� ![]() ��
��
����65%��