��Ŀ����
��ͼ��ʾ����װ�ÿ�������ȡ�۲�Fe(OH)2�ڿ����б���������ɫ�仯��ʵ��ʱ����ʹ����м��6mol/L�����ᣬ�����Լ���ѡ����д���пհף�
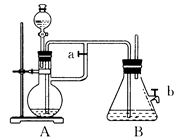
��1��B��ʢ�� ��Һ��A��ӦԤ�ȼ����ҩƷ��________��A�з�Ӧ�����ӷ���ʽ�ǣ�_________________��
��2��ʵ�鿪ʼʱӦ�Ƚ�����a________(����رա�)��
��3����������Fe(OH)2�IJ�������________���������ɵ�Fe(OH)2�����ܽϳ�ʱ�䱣�ְ�ɫ����������___________________________________________��
��4��ʵ����ϣ���b������������һ���ֿ�������ʱBƿ�з�����Ӧ�Ļ�ѧ����ʽΪ��____________________������ ��

��1��B��ʢ�� ��Һ��A��ӦԤ�ȼ����ҩƷ��________��A�з�Ӧ�����ӷ���ʽ�ǣ�_________________��
��2��ʵ�鿪ʼʱӦ�Ƚ�����a________(����رա�)��
��3����������Fe(OH)2�IJ�������________���������ɵ�Fe(OH)2�����ܽϳ�ʱ�䱣�ְ�ɫ����������___________________________________________��
��4��ʵ����ϣ���b������������һ���ֿ�������ʱBƿ�з�����Ӧ�Ļ�ѧ����ʽΪ��____________________������ ��
��1��NaOH;���Fe+2H+=Fe2++H2�� ��2����
��3���رջ���a����A�в�����������A��FeSO4��Һѹ��B�з�����Ӧ:A��Һ�����ص�������������B�У�������ɫ����
��4��4Fe(OH)2+O2+2H2O=4Fe(OH)3��ɫ������ɻ���ɫ������ɺ��ɫ��
��3���رջ���a����A�в�����������A��FeSO4��Һѹ��B�з�����Ӧ:A��Һ�����ص�������������B�У�������ɫ����
��4��4Fe(OH)2+O2+2H2O=4Fe(OH)3��ɫ������ɻ���ɫ������ɺ��ɫ��
�������������a,������ƿA�м���Fe�ۣ�ͨ����Һ©������ƿA�еμ��ӷ��Ե�ϡ���ᣬ��ʱ������Ӧ��Fe+2H+=Fe2++H2������������������ƿB����NaOH��Һ��Ŀ������ϵ�����B�д��������Ļ����С��رջ���b���ٹرջ���a, ����A�в��ϲ���������ʹ��A�е�ѹǿԽ��Խ����ʱA�в�����������Aװ���е�FeSO4��Һͨ��������ѹ��B�У�������ӦFe2++2OH-=Fe(OH)2��.��������װ�ö�����H2�Ļ�ԭ�Ի����У����Կ��Խϳ�ʱ��Ĺ۲��Fe(OH)2�İ�ɫ��������ʵ����ϣ���b���������������뵽װ��B�У���ʱBƿ�з�����Ӧ�Ļ�ѧ����ʽΪ��4Fe(OH)2+O2+2H2O=4Fe(OH)3���ῴ������ɫ������ɻ���ɫ������ɺ��ɫ��˵��Fe(OH)2���ȶ������ױ������е���������Ϊ���ɫ��Fe(OH)3��2��ʵ�����Ʒ������������е������������̵�����Ĺ۲��֪ʶ��

��ϰ��ϵ�д�
�����Ŀ


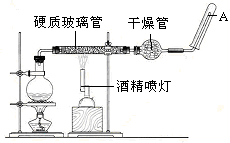
 ����ԭ��Mn2+����Ӧ�����Һ�м�������п�ۣ�����ɫ�պ���ʧ��������Һ�ռ�����ƿ�У���ʱ��Һ�Գ����ԡ�
����ԭ��Mn2+����Ӧ�����Һ�м�������п�ۣ�����ɫ�պ���ʧ��������Һ�ռ�����ƿ�У���ʱ��Һ�Գ����ԡ�