��Ŀ����
[��ѧ-ѡ��3�����ʽṹ������]
������Ҫ�İ뵼����ϣ��������ִ����ӹ�ҵ�Ļ�������ش��������⣺
��1����̬Siԭ���У�����ռ�ݵ�����ܲ����Ϊ
��2������Ҫ�Թ����Ρ�
��3�����ʹ��������ʯ�ṹ���Ƶľ��壬����ԭ����ԭ��֮����
��4�����ʹ��ͨ�����飨SiH4���ֽⷴӦ���Ʊ�����ҵ�ϲ���Mg2Si��NH4Cl��Һ�������з�Ӧ�Ƶ�SiH4���÷�Ӧ�Ļ�ѧ����ʽΪ
��5��̼����йػ�ѧ������������ʾ����Ҫ�����ͽ��������й���ʵ��
�ٹ���̼ͬ�壬Ҳ��ϵ���⻯�������������������϶�Զ���������࣬ԭ����
��SiH4���ȶ���С��CH4���������������ԭ����
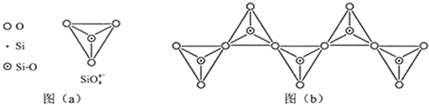
��6���ڹ������У�SiO
�����壨����ͼ��a����ͨ�����ö��������ӿ��γɵ�״����״����״���Ǽ���״�Ĵ���ṹ��ʽ��ͼ��b��Ϊһ�����������ṹ�Ķ�����������Siԭ�ӵ��ӻ���ʽΪ
������Ҫ�İ뵼����ϣ��������ִ����ӹ�ҵ�Ļ�������ش��������⣺
��1����̬Siԭ���У�����ռ�ݵ�����ܲ����Ϊ
M
M
�����ܲ���е�ԭ�ӹ����Ϊ9
9
��������Ϊ4
4
����2������Ҫ�Թ����Ρ�
��������
��������
�Ȼ��������ʽ�����ڵؿ��У���3�����ʹ��������ʯ�ṹ���Ƶľ��壬����ԭ����ԭ��֮����
���ۼ�
���ۼ�
���ϣ��侧���й���8��ԭ�ӣ�����������λ�ù���3
3
��ԭ�ӣ���4�����ʹ��ͨ�����飨SiH4���ֽⷴӦ���Ʊ�����ҵ�ϲ���Mg2Si��NH4Cl��Һ�������з�Ӧ�Ƶ�SiH4���÷�Ӧ�Ļ�ѧ����ʽΪ
Mg2Si+4NH4Cl=SiH4+4NH3+2MgCl2
Mg2Si+4NH4Cl=SiH4+4NH3+2MgCl2
����5��̼����йػ�ѧ������������ʾ����Ҫ�����ͽ��������й���ʵ��
| ��ѧ�� | C-C | C-H | C-O | Si-Si | Si-H | Si-O |
| ����/��kJ?mol-1�� | 356 | 413 | 336 | 226 | 318 | 452 |
C-C����C-H����ǿ�����γɵ������ȶ�����������Si-Si����Si-H���ļ��ܽϵͣ����ѣ����³��������������ɣ�
C-C����C-H����ǿ�����γɵ������ȶ�����������Si-Si����Si-H���ļ��ܽϵͣ����ѣ����³��������������ɣ�
����SiH4���ȶ���С��CH4���������������ԭ����
C-H���ļ��ܴ���C-O����C-H����C-O���ȶ�����Si-H���ļ���ȴԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O��
C-H���ļ��ܴ���C-O����C-H����C-O���ȶ�����Si-H���ļ���ȴԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O��
����6���ڹ������У�SiO
4- 4 |
sp3
sp3
��Si��O��ԭ����֮��Ϊ1��3
1��3
����ѧʽΪSiO32-
SiO32-
��
��������1����ԭ�Ӻ�����14�����ӣ����̬ԭ�ӵĺ�������Ų�ʽΪ1s22s22p63s23p2 ����Ӧ�ܲ�ֱ��ΪK��L��M������������ߵ��������M�㣬���ܲ���s��p��d�����ܼ���s�ܼ���1�������p�ܼ���3�������d�ܼ���5����������Թ���9��ԭ�ӹ������ԭ�ӵ�M�ܲ���4�����ӣ�3s23p2����
��2����Ԫ������Ȼ������Ҫ�Ի���̬��������������Σ���ʽ���ڣ�
��3���辧��ͽ��ʯ�������ƶ�����ԭ�Ӿ��壬��ԭ��֮���Թ��ۼ���ϣ��ڽ��ʯ����ľ����У�ÿ��������һ��̼ԭ�ӣ���������ƽṹ����������λ�ù���ԭ��Ϊ 6��
=3����
��4��Mg2Si��NH4Cl��Һ�������з�Ӧ�Ƶ�SiH4��NH3��MgCl2��
��5���������е�C-C����C-H�����ڹ����е�Si-Si����Si-H���ļ��ܣ�
�ڼ���Խ�����ʾ�Խ�ȶ���C-H���ļ��ܴ���C-O������C-H����C-O���ȶ�����Si-H���ļ���ԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O����
��6���������еĹ������SiO
��Ϊ��������ṹ����������ԭ��Siԭ�Ӳ�ȡ��sp3�ӻ���ʽ������ͼ��b����һ���ṹ��Ԫ�к���1���衢3����ԭ�ӣ���ѧʽΪSiO32-��
��2����Ԫ������Ȼ������Ҫ�Ի���̬��������������Σ���ʽ���ڣ�
��3���辧��ͽ��ʯ�������ƶ�����ԭ�Ӿ��壬��ԭ��֮���Թ��ۼ���ϣ��ڽ��ʯ����ľ����У�ÿ��������һ��̼ԭ�ӣ���������ƽṹ����������λ�ù���ԭ��Ϊ 6��
| 1 |
| 2 |
��4��Mg2Si��NH4Cl��Һ�������з�Ӧ�Ƶ�SiH4��NH3��MgCl2��
��5���������е�C-C����C-H�����ڹ����е�Si-Si����Si-H���ļ��ܣ�
�ڼ���Խ�����ʾ�Խ�ȶ���C-H���ļ��ܴ���C-O������C-H����C-O���ȶ�����Si-H���ļ���ԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O����
��6���������еĹ������SiO
4- 4 |
����⣺��1����ԭ�Ӻ�����14�����ӣ����̬ԭ�ӵĺ�������Ų�ʽΪ1s22s22p63s23p2 ����Ӧ�ܲ�ֱ��ΪK��L��M������������ߵ��������M�㣬���ܲ���s��p��d�����ܼ���s�ܼ���1�������p�ܼ���3�������d�ܼ���5����������Թ���9��ԭ�ӹ������ԭ�ӵ�M�ܲ���4�����ӣ�3s23p2����
�ʴ�Ϊ��M��9��4��
��2����Ԫ������Ȼ������Ҫ�Ի���̬��������������Σ���ʽ���ڣ�
�ʴ�Ϊ����������
��3���辧��ͽ��ʯ�������ƶ�����ԭ�Ӿ��壬��ԭ��֮���Թ��ۼ���ϣ��ڽ��ʯ����ľ����У�ÿ��������һ��̼ԭ�ӣ���������ƽṹ����������λ�ù���ԭ��Ϊ 6��
=3����
�ʴ�Ϊ�����ۼ���3��
��4��Mg2Si��NH4Cl��Һ�������з�Ӧ�Ƶ�SiH4��NH3��MgCl2������ʽΪ��Mg2Si+4NH4Cl=SiH4+4NH3+2MgCl2���ʴ�Ϊ��Mg2Si+4NH4Cl=SiH4+4NH3+2MgCl2��
��5���������е�C-C����C-H�����ڹ����е�Si-Si����Si-H���ļ��ܣ����Թ�����Si-Si����Si-H���ļ������ѣ����³��������������ɣ�
�ʴ�Ϊ��C-C����C-H����ǿ�����γɵ������ȶ�����������Si-Si����Si-H���ļ��ܽϵͣ����ѣ����³��������������ɣ�
�ڼ���Խ�����ʾ�Խ�ȶ���C-H���ļ��ܴ���C-O������C-H����C-O���ȶ�����Si-H���ļ���ԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O����
�ʴ𰸣�C-H���ļ��ܴ���C-O����C-H����C-O���ȶ�����Si-H���ļ���ȴԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O����
��6���������еĹ������SiO
��Ϊ��������ṹ����������ԭ��Siԭ�Ӳ�ȡ��sp3�ӻ���ʽ��
�ʴ�Ϊ��sp3
����ͼ��b����һ���ṹ��Ԫ�к���1���衢3����ԭ�ӣ���ѧʽΪSiO32-��
�ʴ�Ϊ��1��3��SiO32-��
�ʴ�Ϊ��M��9��4��
��2����Ԫ������Ȼ������Ҫ�Ի���̬��������������Σ���ʽ���ڣ�
�ʴ�Ϊ����������
��3���辧��ͽ��ʯ�������ƶ�����ԭ�Ӿ��壬��ԭ��֮���Թ��ۼ���ϣ��ڽ��ʯ����ľ����У�ÿ��������һ��̼ԭ�ӣ���������ƽṹ����������λ�ù���ԭ��Ϊ 6��
| 1 |
| 2 |
�ʴ�Ϊ�����ۼ���3��
��4��Mg2Si��NH4Cl��Һ�������з�Ӧ�Ƶ�SiH4��NH3��MgCl2������ʽΪ��Mg2Si+4NH4Cl=SiH4+4NH3+2MgCl2���ʴ�Ϊ��Mg2Si+4NH4Cl=SiH4+4NH3+2MgCl2��
��5���������е�C-C����C-H�����ڹ����е�Si-Si����Si-H���ļ��ܣ����Թ�����Si-Si����Si-H���ļ������ѣ����³��������������ɣ�
�ʴ�Ϊ��C-C����C-H����ǿ�����γɵ������ȶ�����������Si-Si����Si-H���ļ��ܽϵͣ����ѣ����³��������������ɣ�
�ڼ���Խ�����ʾ�Խ�ȶ���C-H���ļ��ܴ���C-O������C-H����C-O���ȶ�����Si-H���ļ���ԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O����
�ʴ𰸣�C-H���ļ��ܴ���C-O����C-H����C-O���ȶ�����Si-H���ļ���ȴԶС��Si-O��������Si-H�����ȶ����������γ��ȶ��Ը�ǿ��Si-O����
��6���������еĹ������SiO
4- 4 |
�ʴ�Ϊ��sp3
����ͼ��b����һ���ṹ��Ԫ�к���1���衢3����ԭ�ӣ���ѧʽΪSiO32-��
�ʴ�Ϊ��1��3��SiO32-��
������������Ҫ�����˻�̬ԭ�ӵĺ�������Ų�������ṹ����ѧ����ʽ����д���ӻ��������ѧ����֪ʶ���Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
 ��2012?������ģ��[��ѧ--ѡ��3�����ʽṹ������]�飨As����һ����Ҫ�Ļ�ѧԪ�أ�����γɶ�����;�㷺�Ļ����
��2012?������ģ��[��ѧ--ѡ��3�����ʽṹ������]�飨As����һ����Ҫ�Ļ�ѧԪ�أ�����γɶ�����;�㷺�Ļ���� ����ѧ--ѡ��3�����ʽṹ�����ʡ�
����ѧ--ѡ��3�����ʽṹ�����ʡ�