��Ŀ����
18����ҵ����Cl2�ͼ���Ư�ۡ�Ư�۾���Ư��Һ����ش���������⣺��1����ҵ�Ͻ������Ƴ�Ư�۾�����ҪĿ���DZ��ڱ�������䣬��Ư�۾�����Ҫ��Ӧ�Ļ�ѧ����ʽ��2Cl2+2Ca��OH��2=CaCl2+Ca��ClO��2+2H2O��
��2����������¶���ڿ����л���ʣ��ڱ��ʹ����м��з�������ԭ��Ӧ����������������ԭ��Ӧ��������B��
A����ʯ�ҡ� B��Ư�ۡ� C��NaOH�� D��Na2SO3
��3��д������������ѧ����ʽ����ע����Ӧ���ͣ�
Ư��+ϡ���Ca��ClO��2+2HCl=CaCl2+2HClO�������ֽⷴӦ��Ӧ��
Ư��+Ũ���Ca��ClO��2+4HCl��Ũ��=CaCl2+2Cl2��+2H2O����������ԭ��Ӧ��
��4��������ܾõ�Ư����Ũ���ᷴӦ�Ƶõ������У����ܺ��е����������Ǣ٢ۢܣ�����ţ���
��HCl�� ��O2�� ��CO2�� ��H2O������
���� ��1�����������壬���㱣������䣻�������������Ʒ�Ӧ�����Ȼ��ơ�������ƺ�ˮ��
��2���е���ת�ƵĻ�ѧ��Ӧ��������ԭ��Ӧ��
��3��������ƺ�ϡ���ᷢ�����ֽⷴӦ����Ũ���ᷢ��������ԭ��Ӧ��
��4��Ũ�����ӷ�����������ܹ�������еĶ�����̼��ˮ��Ӧ����̼��ƺʹ����ᣬ������ֽ������Ȼ����������������̼��Ʒ�Ӧ���ɶ�����̼���壬�ݴ˽��
��� �⣺��1�����������壬���㱣������䣬������ת��Ϊ���ȶ��������ʱ��ڱ�������䣬���Թ�ҵ�Ͻ������Ƴ�Ư�۾���
��ҵ�Ͻ�����ͨ��ʯ����[Ca��OH��2]��ȡƯ�۾�����ѧ��Ӧ����ʽΪ��2Cl2+2Ca��OH��2=CaCl2+Ca��ClO��2+2H2O���ʴ�Ϊ�����ڱ�������䣬2Cl2+2Ca��OH��2=CaCl2+Ca��ClO��2+2H2O��
��2��A����ʯ���ڿ����о��ñ��ʣ�����CaO+H2O�TCa��OH��2��Ca��OH��2+CO2�TCaCO3��+H2O�������ڷ�������ԭ��Ӧ����A����
B��Ư�ۿ����о��ã��ȷ���CO2+H2O+Ca��ClO��2�TCaCO3+2HClO�����ڷ�������ԭ��Ӧ������2HClO$\frac{\underline{\;����\;}}{\;}$2HCl+O2��������������ԭ��Ӧ����B��ȷ��
C���������ƺͶ�����̼������Ӧ2NaOH+CO2�TNa2CO3+H2O�����ڷ�������ԭ��Ӧ����C����
D�����������ױ������������������ƣ���Ӧ����ʽΪ��Na2SO3+O2=Na2SO4������������ԭ��Ӧ����D����
��ѡB��
��3��Ư�ۺ�ϡ���ᷢ���Ļ�ѧ��Ӧ����ʽΪ��Ca��ClO��2+2HCl=CaCl2+2HClO�����ڸ��ֽⷴӦ��
Ư�ۺ�Ũ���ᷢ���Ļ�ѧ��ӦΪ��Ca��ClO��2+4HCl��Ũ��=CaCl2+Cl2��+2H2O���÷�Ӧ����������ԭ��Ӧ��
�ʴ�Ϊ��Ca��ClO��2+2HCl=CaCl2+2HClO�����ֽⷴӦ��Ca��ClO��2+4HCl��Ũ��=CaCl2+2Cl2��+2H2O��������ԭ��Ӧ��
��4����������ܹ�������еĶ�����̼��ˮ��Ӧ����̼��ƺʹ����ᣬ������ֽ������Ȼ��������������ܾõ�Ư�ۿ��ܴ���̼��ƣ�����ܾõ�Ư�ۿ��ܴ���̼��ƣ������ᷢ��Ca��ClO��2+4HCl��Ũ��=CaCl2+2Cl2��+2H2O��CaCO3+2HCl=CaCl2+CO2��+H2O��
Ũ�����ӷ�������Һ�����ɵ������л����ˮ���������Ƶõ������У����ܺ�����������ΪCO2��HCl��H2O��������
��ѡ���٢ۢܣ�
���� ���⿼�������Ļ�ѧ���ʣ��ӷ�Ӧԭ�����������������ʣ�ע��Ư�ۺ�Ũϡ���ᷴӦ������Ϊ�״��㣮

 ������״Ԫ���Ծ�ϵ�д�
������״Ԫ���Ծ�ϵ�д�| A�� | 12C��14CΪ��ͬ���� | |
| B�� | ʯī��C60�Ļ�Ϊͬ�������� | |
| C�� | H2O��D2O��Ϊͬλ�� | |
| D�� | ����������о������Ȼ���-COOH���Ͱ�����-NH2�� |
| A�� | 7.2 g CaO2�����������Ӻ�����������Ϊ0.3 NA | |
| B�� | �ڱ���£�22.4LHF��22.4LC2H4ԭ�Ӹ�����Ϊ1��3 | |
| C�� | ��1 mol NH4NO3����ϡ��ˮ��ʹ��Һ�����ԣ���Һ��NH4+��ĿΪNA | |
| D�� | 0.1 mol H2O2�����к����Թ��ۼ���ĿΪ0.3 NA |
| A | B | C | D |
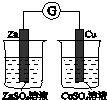 | 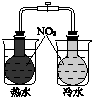 | 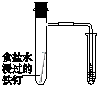 | 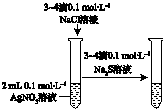 |
| ��֤��ѧ��ת��Ϊ���� | ֤���¶ȶ�ƽ���ƶ���Ӱ�� | ��֤���������ⸯʴ | ��֤AgCl�ܽ��С��Ag2S |
| A�� | A | B�� | B | C�� | C | D�� | D |
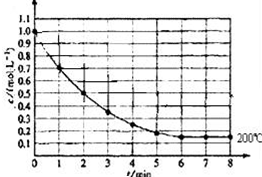 һ�̶�������ܱ������з���1molX��g��������Ӧ��X��g��?4Y��s��+Z��g������ͼ��ʾ�� 200��ʱ��X��Ũ����ʱ��仯�����ߣ�
һ�̶�������ܱ������з���1molX��g��������Ӧ��X��g��?4Y��s��+Z��g������ͼ��ʾ�� 200��ʱ��X��Ũ����ʱ��仯�����ߣ�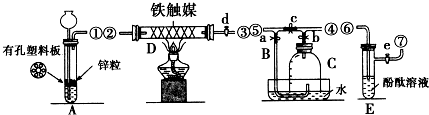
 ��������������Ӧˮ����ļ��Ա�Mg��OH��2ǿ���ǿ������������
��������������Ӧˮ����ļ��Ա�Mg��OH��2ǿ���ǿ������������ ��
�� ��C�Ļ�ѧʽΪC15H13NO4��-Bn����������������
��C�Ļ�ѧʽΪC15H13NO4��-Bn����������������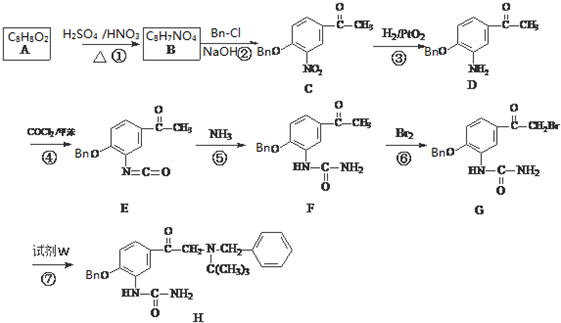
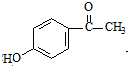 W
W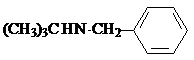 ��
��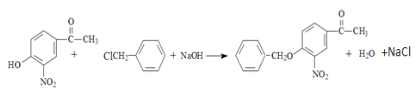 ��
��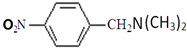 �����Լ����ã�
�����Լ����ã�