��Ŀ����
��ҵ�ϵ�ⱥ��ʳ��ˮǰ����Ҫ��ʳ�ν��о��ƣ���֪��ҵʳ���к���Ca2+��Mg2+��SO42-�����ʣ������ѹ�ҵʳ�ξ����ɾ��Σ���ش��������⣺
��1������ȥSO42-�����������Եı��Լ������Լ��������� ��
A��Ba��OH��2����B��Ba��NO3��2����C��BaCl2
��ѡ���Լ��������� ��
��2����ѡ�õ��Լ���Na2CO3��Ba��OH��2��HCl������Ϊ����ĺ���˳���� ��
��3����ѡ�õ��Լ���BaCl2��NaOH��Na2CO3��HCl�����м����Լ���˳��������� ��
A��BaCl2��NaOH��Na2CO3��HCl
B��NaOH��BaCl2��Na2CO3��HCl
C��Na2CO3��NaOH��BaCl2��HCl
��4����ѡ�ò�ͬ���Լ��Ͳ�ͬ�ļ���˳��ʱ���������������ᣮ����Ϊ��������������ǣ������ֻ����ӷ���ʽ��ʾ�����ԣ� ��
��1������ȥSO42-�����������Եı��Լ������Լ���������
A��Ba��OH��2����B��Ba��NO3��2����C��BaCl2
��ѡ���Լ���������
��2����ѡ�õ��Լ���Na2CO3��Ba��OH��2��HCl������Ϊ����ĺ���˳����
��3����ѡ�õ��Լ���BaCl2��NaOH��Na2CO3��HCl�����м����Լ���˳���������
A��BaCl2��NaOH��Na2CO3��HCl
B��NaOH��BaCl2��Na2CO3��HCl
C��Na2CO3��NaOH��BaCl2��HCl
��4����ѡ�ò�ͬ���Լ��Ͳ�ͬ�ļ���˳��ʱ���������������ᣮ����Ϊ��������������ǣ������ֻ����ӷ���ʽ��ʾ�����ԣ�
���㣺���ʵļ���ͼ���Ļ�������ѡ��Ӧ��
ר�⣺���ʼ��������
��������1������������뱵���ӷ�Ӧ���ɳ������ҳ��Ӳ������������ʣ�
��2������Ca2+��Mg2+��SO42-�����ʣ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ��
��3������Ca2+��Mg2+��SO42-�����ʣ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ��
��4���������������ᣬ�ɳ�ȥ������̼���Ʋ�����pH��
��2������Ca2+��Mg2+��SO42-�����ʣ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ��
��3������Ca2+��Mg2+��SO42-�����ʣ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ��
��4���������������ᣬ�ɳ�ȥ������̼���Ʋ�����pH��
���
�⣺��1������ȥSO42-�����������Եı��Լ�������ѡ��B�������Ba��NO3��2����������NO3-��������Ӧ���Գ�ȥ��
�ʴ�Ϊ��B������Ba��NO3��2����������NO3-����
��2������Ca2+��Mg2+��SO42-�����ʣ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ�����Լ��ļ���˳��ΪBa��OH��2��Na2CO3��HCl��
�ʴ�Ϊ��Ba��OH��2��Na2CO3��HCl��
��3��Mg2+��SO42-�ij���û���Ⱥ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ��������Լ���˳���������AB���ʴ�Ϊ��AB��
��4���������������ᣬ������Ϊ������Һ��pH����ȥ��Һ�е�����CO32-��OH-��CO32-+2H+�TH2O+CO2����OH-+H+�TH2O��
�ʴ�Ϊ��������Һ��pH����ȥ��Һ�е�����CO32-��OH-��CO32-+2H+�TH2O+CO2����OH-+H+�TH2O��
�ʴ�Ϊ��B������Ba��NO3��2����������NO3-����
��2������Ca2+��Mg2+��SO42-�����ʣ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ�����Լ��ļ���˳��ΪBa��OH��2��Na2CO3��HCl��
�ʴ�Ϊ��Ba��OH��2��Na2CO3��HCl��
��3��Mg2+��SO42-�ij���û���Ⱥ�����ȥ�����ӣ���̼����Ӧ����������֮����HCl֮ǰ��������Լ���˳���������AB���ʴ�Ϊ��AB��
��4���������������ᣬ������Ϊ������Һ��pH����ȥ��Һ�е�����CO32-��OH-��CO32-+2H+�TH2O+CO2����OH-+H+�TH2O��
�ʴ�Ϊ��������Һ��pH����ȥ��Һ�е�����CO32-��OH-��CO32-+2H+�TH2O+CO2����OH-+H+�TH2O��
���������⿼�����ʵķ����ᴿ�����ӵȣ�Ϊ��Ƶ���㣬���ճ������Լ��ļ���˳��̼��������������֮����HCl֮ǰΪ���Ĺؼ������ش����ᴿ�Ŀ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
���������ᷴӦ��������NaOH��Һ��Ӧ���ǣ�������
��Na2SiO3 ��Al��OH��3 ��NaHCO3 ��Al2O3��
��Na2SiO3 ��Al��OH��3 ��NaHCO3 ��Al2O3��
| A���٢ڢ� | B���ڢۢ� |
| C���ڢ� | D��ȫ�� |
����˵���У���ȷ���ǣ�������
| A��ȩ�������õ�����һԪ�� |
| B����ȩ����ȩ����ȩ��û��ͬ���칹�� |
| C����2%��ϡ��ˮ��μ���2%��AgNO3��Һ�У�������ǡ���ܽ�Ϊֹ�����Ƶ�������Һ |
| D����2%��NaOH��Һ4��6�Σ�����2mL 10%��CuSO4��Һ���Ƶ�Cu��OH��2����Һ����������ȩ�����Լ� |
����˵������ȷ���ǣ�������
| A�������Ȼ��ܵ����ʣ��ں��в�ͬ��Ŀ�Ľᾧˮ����ʱ���ֲ�ͬ����ɫ������ʼ���ˮ���У��Ƴɱ�ɫˮ�� |
| B���մɾ��п�����������ʴ�����¡���Ե���׳��͵��ŵ� |
| C������û�й̶����۵㣬������״̬ʱ�����Ա����Ƴ��κ���״����Ʒ |
| D�����ά����Ҫ�ɷ־��ǹ赥�ʣ����Ƿdz��õ�ͨ�Ų��� |
������̬�⻯�����ȶ����ǣ�������
| A��HF |
| B��HCl |
| C��HBr |
| D��H2S |
 ��
��
 ��
��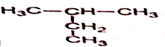
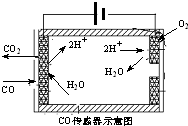 �������ִ������а�����Խ��Խ��Ҫ�Ľ�ɫ������β����̼�⻯����������Pһ����̼�ȣ������Ļ�����ȾԽ��Խ���ԣ������������ŷ��ѳ�Ϊ���д�����Ⱦ����Ҫ��Դ��
�������ִ������а�����Խ��Խ��Ҫ�Ľ�ɫ������β����̼�⻯����������Pһ����̼�ȣ������Ļ�����ȾԽ��Խ���ԣ������������ŷ��ѳ�Ϊ���д�����Ⱦ����Ҫ��Դ��