��Ŀ����
��10.0���2´105Pa�������£���ӦaA(g)ƒdD(g)+eE(g)����ƽ����ڲ������κ����ʵ���������������ϵ��ѹǿ���¶�ά�ֲ��䣩�������г���ͬѹǿ�·�Ӧ����ƽ��ʱ����D��Ũ�ȣ�ѹǿ/Pa�������������������� 2´105������������������������ 5´105������������������������ 1´106
D��Ũ��/mol��L-1�������� 0.085������������������������ 0.20���������������������������� 0.44
���ݱ������ݣ��ش��������⣺
��1��ѹǿ��2´105Pa���ӵ�5´105Paʱ��ƽ����_______��Ӧ�����ƶ�����������桱����������_______��
��2��ѹǿ��5´105Pa���ӵ�1´106Paʱ��ƽ����_______��Ӧ�����ƶ�����������桱����������_______��
������
| ��1����,D��Ũ������ı���Ϊ ��2����,D��Ũ������ı���Ϊ
|

 ��У����ϵ�д�
��У����ϵ�д�(15��)
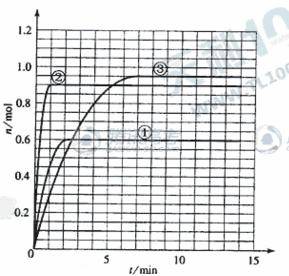
ij�¶�ʱ����2L�ܱ���������̬����X��Y��Ӧ������̬����Z�����ǵ����ʵ�����ʱ��ı仯�����ʾ��
��1�������±������ݣ��ڴ��������Ӧλ���ϻ���X��Y��Z�����ʵ�����n����ʱ�䣨t���仯�����ߣ�
| t/min | X/mol | Y/mol | Z/mol |
| 0 | 1.00 | 1.00 | 0.00 |
| 1 | 0.90 | 0.80 | 0.20 |
| 3 | 0.75 | 0.50 | 0.50 |
| 5 | 0.65 | 0.30 | 0.70 |
| 9 | 0.55 | 0.10 | 0.90 |
| 10 | 0.55 | 0.10 | 0.90 |
| 14 | 0.55 | 0.10 | 0.90 |

(2)��ϵ�з�����Ӧ�Ļ�ѧ����ʽ��___________________________;
(3)��ʽ����÷�Ӧ��0-3minʱ���ڲ���Z��ƽ����Ӧ���ʣ�_______________;
(4)�÷�Ӧ�ﵽƽ��ʱ��Ӧ��X��ת����a����___________________________;
(5)����÷�Ӧ�Ƿ��ȷ�Ӧ���ı�ʵ���������¶ȡ�ѹǿ���������õ�Z��ʱ��仯������1��2��3������ͼ��ʾ��������1��2��3����Ӧ��ʵ�������ı�ֱ��ǣ�
��_________________ ��_______________ ��__________________
 CH3OH��g����H1=-116kJ?mol��1
CH3OH��g����H1=-116kJ?mol��1
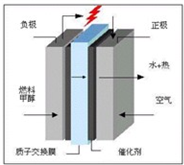 ��������ͼ����ָ��ͼ�е�ѹǿP1=
��������ͼ����ָ��ͼ�е�ѹǿP1= CO��g��+2H2��g����Ӧ��ƽ�ⳣ��Ϊ
CO��g��+2H2��g����Ӧ��ƽ�ⳣ��Ϊ