��Ŀ����
ú����Ҫ����Դ��Ҳ�ǻ�����������Ҫԭ�ϡ�
(1)úȼ�ղ����ķ���ֱ���ŷŵ������У����ܵ��µĻ�����Ⱦ������_______��
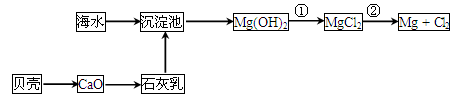
(2)��ͼ�Ƕ�ú��ȼ�ղ����ķ������г���������������ʾ��ͼ��
�ٷ�����������У���Ҫ��Ӧ�Ļ�ѧ����ʽΪ______________��_____________��
����ú����ֱ������һ�����ʣ�����Ч����ú��ȼ�ղ�����SO2����������___��
��ʯ��Ĺ�ҵ�����е���;��_________________(д��һ����;����)��
(3)ú����___________(��ӹ�����)���Եõ���¯ú����ú���ͺͽ�̿��ú���;���_______(��ӹ�����)�ɵõ������廯���ú��Ҳ�������⻯��ת��ȼ�ͣ��⻯���ı�����______________��
(4)ú����Һ��ת��ΪCH3OH��������Ƴ���KOH��Һ���õ缫�����һ��ϸС�IJ������缫�����������������ǿ�������ȶ�������CH3OH��O2���ɵ�ȼ�ϵ�ص�����ת����Ҫ��ʽ������ ��������ӦʽΪ�������� ���缫����Ʋ��۵�ԭ��Ϊ ������
(1)���ꡢ�۳���Ⱦ
(2)��SO2��CaCO3 CaSO3��CO2��2CaSO3��O2��2CaSO4
��CaCO3��CaO��Ca(OH)2
��ҽ�á��������ϡ�ģ�͵Ⱥ�����
(3)����������ú����Ԫ�صú����������ԭ������̼ԭ�����ı�ֵ
(4)�ɻ�ѧ��ת��Ϊ���ܣ�CH3OH+8OH- -6e-=CO32-+6H2O������缫��λ�������CH3OH��O2�����������ٵ缫��Ӧ���ʡ�
����

����Ԫ�آ�Cl����Na����Br����I����Mg����U���ں�ˮ�е���Ԫ�ص���
| A���٢ڢ� | B���ܢ� | C���٢ڢۢ� | D���ۢܢݢ� |
���н����Ĺ�ҵ�Ʒ���ȷ����(����)
| A�����ѣ��ý������û����Ȼ���(TiCl4)��Һ�е��� |
| B���������ý�̿�Ϳ�����Ӧ������һ����̼�ڸ����»�ԭ����ʯ�е��� |
| C�����ƣ��ú�ˮ��ԭ���Ƶþ��Σ��ٵ�ⴿ���Ȼ�����Һ�õ������� |
| D����ͭ���û�ͭ��⾫���õ�����Ϊ99.9%��ͭ |



 �������ѱ䣬�����⣨2H�������˾۱䣬��LiH��Ұ����������ܺ�ˮɹ��
�������ѱ䣬�����⣨2H�������˾۱䣬��LiH��Ұ����������ܺ�ˮɹ��
 Na2S2O5��H2O�ȶಽ��Ӧ��
Na2S2O5��H2O�ȶಽ��Ӧ��