��Ŀ����
��1����ʵ��������С��ʵ�飺������A��B��һ����������һ�������Ϊ ��Բ����������ʹѹǿΪP��Ȼ��������ϵ�ü��������ȵ�280�棬�������·�Ӧ
��Բ����������ʹѹǿΪP��Ȼ��������ϵ�ü��������ȵ�280�棬�������·�Ӧ ����ƽ��ÿ��������0��5 mol��C��������������ɢ������ƽ��Ϊ
����ƽ��ÿ��������0��5 mol��C��������������ɢ������ƽ��Ϊ ��Ϊ��ά�ֺ���280�棬ƽ��ÿ�������ü������������ṩ����kJ���������������������̣�����
��Ϊ��ά�ֺ���280�棬ƽ��ÿ�������ü������������ṩ����kJ���������������������̣�����
��_________________________________________������
��2��������1����С��ʵ������Ϊ��ҵ����������Ӧ�����ĵ���뾶�߶ȶ�����ԭ������10�������ɡ������塱���������н�A��B��ԭ�������룬ʹѹǿ��ΪP��Ȼ����ȵ�280��ʹ��Ӧ��ʼ��Ϊ��ά�ֺ���280�棬��Ӧ��ʼ��Ӧ�������Ȼ��ǽ�����ȴ?������������ɢ�����ʲ��䣩����
ƽ��ÿ�����ü���������ϵ�ṩ��������ȴ�������˶���kJ���������������������̣�����
��________________________________________������
��3���ں��¡���ѹ�£���2���ġ������塱ƽ����ϵ�н�A��B��ԭ�������룬���´ﵽƽ��ʱ��C���������_________��
 ��Բ����������ʹѹǿΪP��Ȼ��������ϵ�ü��������ȵ�280�棬�������·�Ӧ
��Բ����������ʹѹǿΪP��Ȼ��������ϵ�ü��������ȵ�280�棬�������·�Ӧ ����ƽ��ÿ��������0��5 mol��C��������������ɢ������ƽ��Ϊ
����ƽ��ÿ��������0��5 mol��C��������������ɢ������ƽ��Ϊ ��Ϊ��ά�ֺ���280�棬ƽ��ÿ�������ü������������ṩ����kJ���������������������̣�����
��Ϊ��ά�ֺ���280�棬ƽ��ÿ�������ü������������ṩ����kJ���������������������̣�������_________________________________________������
��2��������1����С��ʵ������Ϊ��ҵ����������Ӧ�����ĵ���뾶�߶ȶ�����ԭ������10�������ɡ������塱���������н�A��B��ԭ�������룬ʹѹǿ��ΪP��Ȼ����ȵ�280��ʹ��Ӧ��ʼ��Ϊ��ά�ֺ���280�棬��Ӧ��ʼ��Ӧ�������Ȼ��ǽ�����ȴ?������������ɢ�����ʲ��䣩����
ƽ��ÿ�����ü���������ϵ�ṩ��������ȴ�������˶���kJ���������������������̣�����
��________________________________________������
��3���ں��¡���ѹ�£���2���ġ������塱ƽ����ϵ�н�A��B��ԭ�������룬���´ﵽƽ��ʱ��C���������_________��
��1����1���Ӽƣ�����0��5mol C��������45kJ��������ɢ��
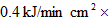
 ������Ӧ�ü�����ÿ�����ṩ����120kJ-45kJ��75 kJ ��
������Ӧ�ü�����ÿ�����ṩ����120kJ-45kJ��75 kJ ��
��2�������뾶�߶ȶ���Ϊԭ��10����������Ϊԭ��1000����ƽ��ÿ��������CΪ
0��5 mol��1000��500 mol���ų�����45000kJ��������ɢ��12000kJ��������ȴ��ÿ������������
45000kJ-12000kJ��33000kJ ����
��3������
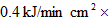
 ������Ӧ�ü�����ÿ�����ṩ����120kJ-45kJ��75 kJ ��
������Ӧ�ü�����ÿ�����ṩ����120kJ-45kJ��75 kJ ����2�������뾶�߶ȶ���Ϊԭ��10����������Ϊԭ��1000����ƽ��ÿ��������CΪ
0��5 mol��1000��500 mol���ų�����45000kJ��������ɢ��12000kJ��������ȴ��ÿ������������
45000kJ-12000kJ��33000kJ ����
��3������

��ϰ��ϵ�д�
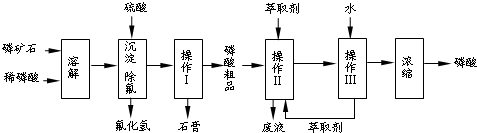
 ����ͼ���������������ϵ�д�
����ͼ���������������ϵ�д�
�����Ŀ
��һ�����ʵ���Ũ����Һ�����ƺ�����к͵ζ�����ѧ��ѧ���������͵Ķ���ʵ�顣ij�о���ѧϰС����ʵ����������1mol/L��ϡ�������Һ��Ȼ������ζ�ijδ֪Ũ�ȵ�NaOH��Һ�������й�˵������ȷ����______________���𰸿��ܲ�Ψһ��
| A��ʵ�������õ��ĵζ��ܡ�����ƿ����ʹ��ǰ����Ҫ��©�� |
| B�����ʵ��������60mL ��ϡ�������Һ������ʱӦѡ��100mL����ƿ�� |
| C������ƿ�к�����������ˮ���ᵼ���������Һ��Ũ��ƫС�� |
| D����ʽ�ζ���������ˮϴ�Ӻ�װ���Ũ�ȵ�ϡ���ᣬ���õ�NaOH��Һ��Ũ�Ƚ�ƫ�� |
F��������Һ���к͵ζ�������ʵ���У��������һ�ζ��������Ӷ���������ʵ������ƫ��
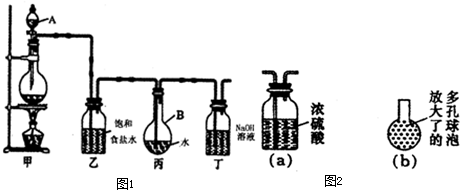
�� . ������ͼ��ʾ��װ����ȡ�϶����ı�����ˮ���ⶨ������ˮ��pH��
�ش��й����⣺
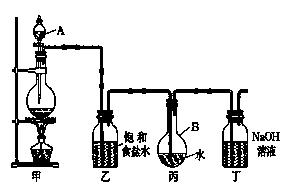
��1��д���йػ�ѧ����ʽ��
װ�üף�____________________________________ ��
װ�ö��� __________________ ��
��2��֤����ˮ�ѱ��͵������� ��
��3����ȡ����ʱ��װ�ñ���Һ���к��е����� ����
 �����ű�ʾ ����
�����ű�ʾ ����(4)����ȥװ���ң�ֱ�ӽ�װ�üͱ���������������ʵ��ⶨ�����Ӱ���ǣ� ��
�ⶨ������ˮ��pH������_______________________________________________��
��.ʵ����ƣ�֤��NaOH�����ڿ����з��ò��ֱ���
_______________________________________________________________
 _______________________________________________________________________
_______________________________________________________________________