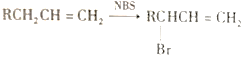��Ŀ����
����Ŀ��I.�Ķ������������������ϣ�
����һ
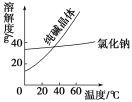
���϶�
���� | �۵�/�� | �е�/�� | �ܶ�/ g/cm3 | �ܽ��� |
�Ҷ���(C2H6O2) | ��11.5 | 198 | 1.11 | ������ˮ���Ҵ� |
������(C3H8O3) | 17.9 | 290 | 1.26 | �ܸ�ˮ���ƾ�������Ȼ��� |
�ش���������(����ĸ���)��
��1���Ӻ�����NaCl��Na2CO3���ᴿNa2CO3�IJ���Ϊ�ܽ⡢_________��________�����ˡ�ϴ�ӡ�����
��2�����Ҷ����ͱ�������������ѷ�����_________��
A��������������B����ȡ�� C���ᾧ�� D����Һ��
II.����������õĵֿ�ű����ҩ��ɴӻƻ��ᆬҶ����ȡ��������ɫ��״���壬�������Ҵ������ѵ��л��ܼ���������ˮ����������ȡ��������
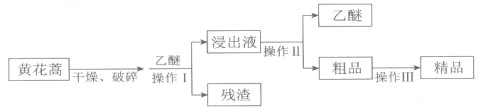
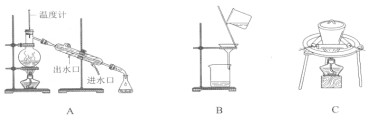
����I��II�У������õ���װ����________������ţ���
��.ʵ������98%���ܶ�Ϊ1.84g/cm3��Ũ��������0.2mol/L��H2SO4��Һ470mL.
��1����Ҫʹ�õ���Ҫ��������Ͳ���ձ�����������___________��__________��
��2�����������ɷֽ�Ϊ���¼�����
A������Ͳ��ȡ______mLŨ���ᣬ����ע��װ��Լ50mL����ˮ���ձ�����ò��������衣
B������������ˮ������ϴ���ձ��Ͳ���������ÿ�ε�ϴҺ����������ƿ�
C����ϡ�ͺ������С�ĵ��ò�������������ƿ�
D���������ƿ�Ƿ�©ˮ��
E��������ˮֱ�Ӽ�������ƿ����Һ��ӽ��̶���1-2cm����
F���ǽ�ƿ���������ߵ���ҡ����Һ��
G���ý�ͷ�ι�������ƿ����μ�������ˮ����Һ����͵�ǡ����������С�
��ݴ���д��
��������������еĿհ״���
�ڲ��������ȷ�IJ���˳������ĸ��д����____________
�� D ������ A ���� �� �� �� ���� F ����
��3�����в�����ʹ����ϡ����Ũ��ƫ�ߵ���______________��
A��ȡ��Ũ����ʱ���ӿ̶���
B������ƿ������ˮϴ�Ӻ���1.2mol/L��������ϴ
C��ת����Һʱ��������������Һ����
D������ʱ��������ƿ�̶���
E����ϡ�ͺ��ϡ��������ת������ƿ�ҽ��к����ʵ�����
F�����ݺ�����ƿ����ҡ�ȣ����ź���Һ����ڿ̶��ߣ��ֲ��伸�� ����ˮ���̶���
���𰸡�����Ũ�� ��ȴ�ᾧ�����½ᾧ�� A C 500ml����ƿ ��ͷ�ι� 5.4ml CBEG ABE
��������
����̼���ƺ��Ȼ��Ƶ��ܽ�����¶ȵı仯���������Ӧ���ýᾧ�����롣���ܵ�Һ���������÷е�����������ķ������롣����װ�õ��ص������AΪ����װ�ã�BΪ����װ�ã�CΪ���ȹ����װ�á�����������Һ�Ĺ��������ʵı仯����Һ������ı仯������������
I. (1)̼���Ƶ��ܽ�������¶ȱ仯�Ӵ������ýᾧ�����룬ʵ�����Ϊ�ܽ⣬����Ũ������ȴ�ᾧ�����½ᾧ�������ˣ�ϴ�ӣ����
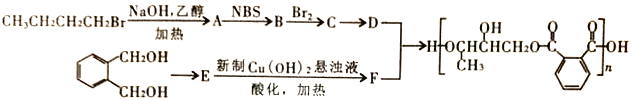
(2)�Ҷ����ͱ��������ܣ����߷е㲻ͬ����������ķ������룬��A��
II.��ȡ�������й��˺������ʵ����̣����漰���ȹ��壬����ѡC��
��. (1)������Һ�����ѡ�� 500ml����ƿ�������Ҫʹ�ý�ͷ�ιܡ�
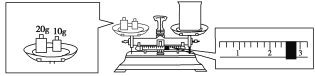
(2) ����Ҫ��Ũ��������Ϊ![]() = 5.4ml��
= 5.4ml��
�� ʵ��ǰ�ȼ������ƿ�Ƿ�©ˮ����ȡһ�������Ũ�������ϡ�ͣ�Ȼ��ϡ�ͺ����������������ƿ�У���ϴ���ձ��Ͳ���������ˮ�����ӽ��̶���1-2cm�����ý�ͷ�ι�������ƿ����μ�������ˮ����Һ����͵�ǡ����������У��ǽ�ƿ���������ߵ���ҡ����Һ�ʲ���˳��Ϊ��CBEG��
(3) A��ȡ��Ũ����ʱ���ӿ̶��ߣ���Ũ��������ƫ��Ũ��ƫ����ȷ��B������ƿ������ˮϴ�Ӻ���1.2mol/L��������ϴ������������࣬Ũ��ƫ��C��ת����Һʱ��������������Һ��������������ʧ��Ũ��ƫС��D������ʱ��������ƿ�̶��ߣ���Һ�����ƫ��Ũ��ƫС��E����ϡ�ͺ��ϡ��������ת������ƿ�ҽ��к����ʵ�������û����ȴ������ȴ����Һ�����ƫС��Ũ��ƫ��F�����ݺ�����ƿ����ҡ�ȣ����ź���Һ����ڿ̶��ߣ��ֲ��伸������ˮ���̶��ߣ���Һ���ƫ��Ũ��ƫС����ѡABE��

 ������ÿ�ʱ��ҵϵ�д�
������ÿ�ʱ��ҵϵ�д�����Ŀ���й�������ŵ����2020�꣬��λGDP������̼�ŷű�2005���½�40%��50%��CO2��ת�����л���ʵ��̼ѭ������Ч����̼�ŷš�
��1�������Ϊ1 L�ĺ����ܱ������У�����1 mol CO2��3 mol H2��һ�������·�Ӧ��CO2(g)��3H2(g)![]() CH3OH(g)��H2O(g)����H����49.0 kJmol��1�����CO2��CH3OH(g)Ũ����ʱ��仯��ͼ��ʾ��
CH3OH(g)��H2O(g)����H����49.0 kJmol��1�����CO2��CH3OH(g)Ũ����ʱ��仯��ͼ��ʾ��
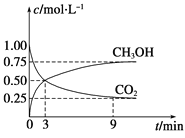
�ټ��㣺��0min��3min��H2��ƽ����Ӧ����v(H2)��_______mol��L��1��min��1����Ӧ��ƽ��ʱ���ų���������_______kJ��
����˵��������Ӧ�ﵽƽ��״̬����________(����)��
A��c(CO2) ��c(CH3OH)��1��1
B�����������ܶȲ��ٷ����仯
C����λʱ��������3mol H2��ͬʱ����1mol H2O
D��CO2����������ڻ�������б��ֲ���
�۷�Ӧ�ﵽƽ����������������䣬�ܼӿ췴Ӧ������ʹ��ϵ����������ʵ������٣��ɲ�ȡ�Ĵ�ʩ��________(����)��
A�������¶� B����С�������
C���ٳ���CO2���� D��ʹ�ú��ʵĴ���
��2����֪A(g)��B(g) ![]() C(g)��D(g) ��H����Ӧ��ƽ�ⳣ�����¶ȵĹ�ϵ���£�
C(g)��D(g) ��H����Ӧ��ƽ�ⳣ�����¶ȵĹ�ϵ���£�
�¶�/ �� | 700 | 800 | 830 | 1000 | 1200 |
ƽ�ⳣ�� | 1.7 | 1.1 | x | 0.6 | 0.4 |
�ش��������⣺
�ٸ÷�Ӧ��_____��Ӧ������ȡ������ȡ�)��
��830��ʱ����һ��2L���ܱ������г���0.20mol��A��0.80mol��B����Ӧ��10sʱ�ﵽƽ�⣬���A��ת����Ϊ80%������830��ʱ����Ӧ��ƽ�ⳣ��x��____��