��Ŀ����
�������岻��ȱ�ٵ���Ԫ�أ����뺬���Ļ�����ɲ��������������ơ����г���һ�ֳ����IJ���ҩƷ���±���˵����IJ������ݡ������ ÿƬ������������ ����Ӧ֢�� ����ȱ����ƶѪ֢��Ԥ���������� �������÷��� ����Ԥ���� �����ء� �ܹ⡢�ܷ⡢�ڸ��ﴦ���� |
(1)��ҩƷ��Fe2+�Ỻ�����������ҹ涨��ҩ����Fe2+�������ʳ���10.00%�������ٷ��á�
��Ϊ�˼���ijҩ����۵ġ������ơ��Ƿ�������ʵ����Ӧѡ�õļ����Լ�Ϊ__________(���Լ�������)��
��ʵ���ҿɲ���H2SO4�ữ��KMnO4��Һ���ԡ������ơ��е�Fe2+���еζ�(����ҩƷ�У������ɷֲ���KMnO4��Ӧ)������ƽ�������ӷ���ʽ��
__________![]() +__________Fe2++__________H+
+__________Fe2++__________H+![]() __________Mn2++__________Fe3++__________H2O
__________Mn2++__________Fe3++__________H2O
�۳�����������Ԫ����������Ϊ20.00%�ġ������ơ�
(2)��֪������������Է�������Ϊ172��������Ϊ�л��ᡣ��
(1)�����軯����Һ
��1��5��8����1��5��4
�۲����� n(Fe2+)=0.01 mol��L-1��12.00��10
m(Fe2+)=0.03 mol��
(
(2)HOOC��CH2��CH2��COOH+2NaOH![]() NaOOC��CH2��CH2��COONa+2H2O
NaOOC��CH2��CH2��COONa+2H2O
FeC4H4O4
������(1)��
KMnO4 �� 5Fe2+
1 5
0.01 mol��L-1��12.00 mol c(Fe2+)��20.00 mol
�ã�c(Fe2+)=0.03 mol��L-1
1 000 mL��Һ�к�Fe2+ 0.03 mol��
��ҩƷ��Fe2+��������Ϊ![]() ��100%=16.00%��10.00%�����Բ����Է���.
��100%=16.00%��10.00%�����Բ����Է���.
(2)��������ΪһԪ��ʱ������Է�������Ϊ![]() =59���������л������ԭ��.��������Ϊ��Ԫ��ʱ������Է�������Ϊ59��2=118���������������������λ�ò�ͬ����ԭ�ӣ�����������Ľṹ��ʽΪHOOC��CH2��CH2��COOH�������������Ļ�ѧʽΪC4H4O4Fe(��FeC4H4O4).
=59���������л������ԭ��.��������Ϊ��Ԫ��ʱ������Է�������Ϊ59��2=118���������������������λ�ò�ͬ����ԭ�ӣ�����������Ľṹ��ʽΪHOOC��CH2��CH2��COOH�������������Ļ�ѧʽΪC4H4O4Fe(��FeC4H4O4).

�������岻��ȱ�ٵ���Ԫ�أ����뺬���Ļ�����ɲ��������������ơ����г���һ�ֳ����IJ���ҩƷ���±���˵����IJ������ݡ���.��.��.Դ.��
| [���]ÿƬ������������ [��Ӧ֢]����ȱ����ƶѪ֢��Ԥ���������á� [�����÷�]����Ԥ���� С������Ԥ���� [����]�ܹ⡢�ܷ⡢�ڸ��ﴦ���档 |
(1) ��ҩƷ��Fe2+�Ỻ�����������ҹ涨��ҩ����Fe2+�������ʣ��Ѿ�������Fe2+��������Fe2+�������ı�ֵ������10.00% �������ٷ��á�
��ʵ���ҿɲ���H2SO4�ữ��KMnO4��Һ���ԡ������ơ��е�Fe2+���еζ�(����ҩƷ�������ɷݲ���KMnO4��Ӧ)����д���÷�Ӧ�����ӷ���ʽ��
��ʵ��ǰ������Ҫ��ȷ����һ�����ʵ���Ũ�ȵ�KMnO4��Һ250 mL������ʱ��Ҫ�IJ������������������ձ�����ͷ�ι��⣬���� ��
��ijͬѧ��������еζ���ʽ���гֲ�����ȥ������������� ��������ĸ��ţ�

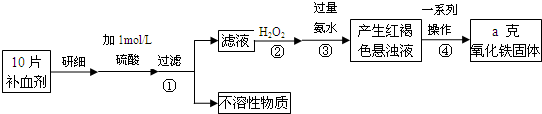
(2) ������������Ԫ����������Ϊ20.00%�ġ������ơ�10.00 g ������ȫ������ϡH2SO4�У����Ƴ�1000 ml��Һ��ȡ��20.00 ml����0.01000 mol•L-1��KMnO4��Һ�ζ�����ȥKMnO4��Һ12.00 ml ����ҩƷ��Fe2+��������Ϊ ��
(3) ��֪������Ϊ��Ԫ�л����ᣬ��23.6 g ���������Һ��4.0 mol•L-1 100.0 ml������������Һǡ����ȫ�к͡��˴Ź�����������ʾ�������������ͼ��ֻ���������շ塣д����������Һ������������Һ��ȫ�к͵Ļ�ѧ����ʽ(�л���д�ṹ��ʽ)
��