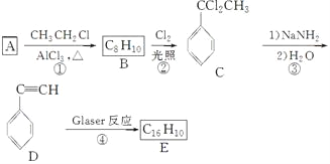
��Ŀ����
����Ŀ���ṹ��ѧ��ԭ�ӡ�����ˮƽ�ϰ���������ʶ���ʹ��ɵĹ��ɣ�����֮�䲻ͬ��������Ϊ�������о���ͬ�������ʵ��й����ʣ������ʽṹ�������ʵ��ӽ�Ԥ�����ʵ��й����ʡ�
��1������˵����ȷ���� ______������ĸ����
A��Ԫ�ص������ɴ�С��˳��O>N>C
B��һ�����أ�CO(NH2)2�������к�1���м���7���Ҽ�
C���Ȼ��ƺ��Ȼ�菉��������ͬ�ľ���
��2�����ݵȵ�����ԭ�����ʻ���OCS�����ӵĵ���ʽӦΪ______��������COCl2�������ڸ�ԭ������㶼����8�����ȶ��ṹ����������ӵĿռ乹��Ϊ____����������������
��3��Cu2+��̬�ĵ����Ų�ʽΪ______��������ͭ��Һ����Ũ�����½ᾧ��������ɫ���壬ȡ25.0g����������������һ���¶������գ����ʣ���������ʼ�ձ���Ϊ23.2g����һ�����������¶ȣ������Ϊ��ɫ������Ϊ16.0g����֪�þ������������������ڽ�ṹ�Ƚ��ȶ������Ʋ�������Ļ�ѧʽ���Ա�ʾΪ______��
��4��ͨ������£���Һ�е�Fe3+��Fe2+�ȶ���������______��
���𰸡�B![]() ƽ��������[Ar]3d9[Cu(H2O)4]SO4��H2OFe3+����Χ�����Ų�Ϊ3d5���ﵽ������ȶ��ṹ��Fe2+ ����Χ�����Ų�Ϊ3d6��δ�ﵽ������ȶ��ṹ
ƽ��������[Ar]3d9[Cu(H2O)4]SO4��H2OFe3+����Χ�����Ų�Ϊ3d5���ﵽ������ȶ��ṹ��Fe2+ ����Χ�����Ų�Ϊ3d6��δ�ﵽ������ȶ��ṹ
��������
��1��A���ڢ�A��Ĵ��ڵ���A��ģ��������һ�����ܴ�С˳����N��O��B�� CO��NH2��2��Cԭ������ԭ��֮���γ�C=O˫����Cԭ����Nԭ��֮���γ�2��C-N������Nԭ����Hԭ��֮�乲�γ�4��N-H����C���Ȼ��Ϊ��������������������İ˸�������,������ģ����Ȼ���Ϊ�����������Ⱥ�����������Ķ��㡢���ġ�������������У���������ͬ��
��2���ʻ���OCS����CO2Ϊ�ȵ����壬����CO2���ӽṹ��������COCl2����ԭ������㶼����8�����ȶ��ṹ��Cԭ������ԭ��֮���γ�C-Cl������Cԭ����Oԭ��֮���γ�C=O˫�����������ӵĽṹʽ��![]() ��������Cԭ�Ӳ�ȡsp2�ӻ��ɼ���
��������Cԭ�Ӳ�ȡsp2�ӻ��ɼ���
��3������Cu�ĵ����Ų�ʽ��дCu2+���ӵĵ����Ų�ʽ�����ݾ��������仯�����жϷ�����
��4��Fe3+����Χ�����Ų�Ϊ3d5���ﵽ������ȶ��ṹ��Fe2+ ����Χ�����Ų�Ϊ3d6��δ�ﵽ������ȶ��ṹ��
��1��A��C��N��O����ͬһ����Ԫ����ԭ��������������ͬһ����Ԫ�صĵ�һ�����ܳ��������ƣ����ڢ�A�嵪ԭ�ӵ�2p������������һ�����ܴ��ڵ���A���Oԭ�ӣ��������һ�����ܴ�С˳����N��O��C��ѡ��A����B��CO��NH2��2��Cԭ������ԭ��֮���γ�C=O˫����Cԭ����Nԭ��֮���γ�2��C-N������Nԭ����Hԭ��֮�乲�γ�4��N-H����1�����ط���[CO��NH2��2]�к�1���м���7���Ҽ���ѡ��B��ȷ��C���Ȼ��Ϊ��������������������İ˸�������,������ģ����Ȼ���Ϊ�����������Ⱥ�����������Ķ��㡢���ġ�������������У���������ͬ��ѡ��C����ѡB��
��2���ʻ���OCS����ԭ������㶼����8�����ȶ��ṹ��Cԭ����Oԭ��֮���γ�C=O˫����Cԭ����Sԭ��֮���γ�C=S˫��������ʽ��![]() ��������COCl2����ԭ������㶼����8�����ȶ��ṹ��Cԭ������ԭ��֮���γ�C-Cl������Cԭ����Oԭ��֮���γ�C=O˫�����������ӵĽṹʽ��
��������COCl2����ԭ������㶼����8�����ȶ��ṹ��Cԭ������ԭ��֮���γ�C-Cl������Cԭ����Oԭ��֮���γ�C=O˫�����������ӵĽṹʽ��![]() ��̼ԭ�ӳ�3���Ҽ����ӻ������ĿΪ3��������Cԭ�Ӳ�ȡsp2�ӻ��ɼ�����COCl2Ϊƽ�������Σ�
��̼ԭ�ӳ�3���Ҽ����ӻ������ĿΪ3��������Cԭ�Ӳ�ȡsp2�ӻ��ɼ�����COCl2Ϊƽ�������Σ�
��3��Cu��29��Ԫ�أ�ԭ�Ӻ��������Ϊ29����̬ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d104s1��ͭԭ��ʧȥ4s��3d�ϸ�һ�������γ�Cu2+����Cu2+���ӵĵ����Ų�ʽΪ��1s22s22p63s23p63d9��[Ar]3d9��ȡ25.0g���壨0.1molCuSO45H2O��������������һ���¶������գ����ʣ���������ʼ�ձ���Ϊ23.2g(����0.1mol H2O)����һ�����������¶ȣ������Ϊ��ɫ������Ϊ16.0g(�ټ���0.4mol H2O��Ϊ0.1mol CuSO4)���Ʋ�������Ļ�ѧʽ���Ա�ʾΪ[Cu(H2O)4]SO4��H2O��
��4��ͨ������£���Һ�е�Fe3+��Fe2+�ȶ���������Fe3+����Χ�����Ų�Ϊ3d5���ﵽ������ȶ��ṹ��Fe2+ ����Χ�����Ų�Ϊ3d6��δ�ﵽ������ȶ��ṹ��

 �����ܿ����ϵ�д�
�����ܿ����ϵ�д�����Ŀ���ϳɰ��ķ�ӦΪN2(g)��3H2(g)![]() 2NH3(g)���±���ij�η�ӦʱNH3��Ũ�ȱ仯��
2NH3(g)���±���ij�η�ӦʱNH3��Ũ�ȱ仯��
ʱ��(min) | 0 | 5 | 10 | 15 | 20 | 25 |
c(NH3)(mol��L-1) | 0 | 0.30 | 0.44 | 0.50 | 0.50 | 0.53 |
����˵����ȷ����
A. 0��10 minʱ����H2��ʾ��ƽ����Ӧ����Ϊ0.044mol��L-1��min-1
B. ����ѹǿ�ͽ����¶Ⱦ��ܼӿ�ϳɰ��ķ�Ӧ����
C. ��ͼ����֪���÷�Ӧ��15��20 minʱ���ڻ�ѧƽ��״̬
D. ��Ӧ������������ܶȱ��ֲ��䣬˵���÷�Ӧһ���ﵽƽ��״̬