��Ŀ����
��֪��Ӧ2CH3OH(g) CH3OCH3(g)+H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400�����¶��£�������㶨���ܱ������м���һ������CH3OH����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
CH3OCH3(g)+H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400�����¶��£�������㶨���ܱ������м���һ������CH3OH����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
���� | CH3OH | CH3OCH3 | H2O |
Ũ��/mol��L-1 | 0.44 | 0.6 | 0.6 |
������������ȷ���� ( )
A���÷�Ӧ��ƽ�ⳣ������ʽΪK=[c(CH3OCH3)��c(H2O)]/c(CH3OH)
B����ʱ�����淴Ӧ���ʵĴ�С��v����v��
C������10 min��Ӧ�ﵽƽ�⣬��ʱc(CH3OH)=0.04 mol��L����
D��0��10min��ƽ����Ӧ����v(CH3OH)=1.6 mol��(L��min)����
��ϰ��ϵ�д�
 Сѧ�̲�ȫ��ϵ�д�
Сѧ�̲�ȫ��ϵ�д�
�����Ŀ



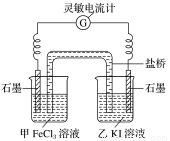
 2Fe2����I2����Ƴ���ͼ��ʾ��ԭ��ء�
2Fe2����I2����Ƴ���ͼ��ʾ��ԭ��ء� CH3COO����H�� ��H1��0 (K1��1.75��10��5)
CH3COO����H�� ��H1��0 (K1��1.75��10��5) 2C(g) ��H��0������������ȷ���� ��
2C(g) ��H��0������������ȷ���� ��  ��
��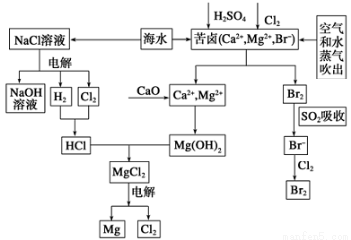
 ��Ca2+��Mg2+��SO42�������ӣ�Ϊ��ȥ��Щ���ӣ������Լ������Ⱥ�˳��Ϊ(д��ѧʽ) ___________________��
��Ca2+��Mg2+��SO42�������ӣ�Ϊ��ȥ��Щ���ӣ������Լ������Ⱥ�˳��Ϊ(д��ѧʽ) ___________________�� l2��6H2O
l2��6H2O MgCl2+6H2O���÷�ӦҪ��HCl�����н��У�ԭ����_______________��
MgCl2+6H2O���÷�ӦҪ��HCl�����н��У�ԭ����_______________��