��Ŀ����
����Ŀ��������ȡ���Һ��Ͻ��еIJ�������![]() ����ȡ�����ӵ�ˮ����ȡ�ǣ�������ǣ� ��
����ȡ�����ӵ�ˮ����ȡ�ǣ�������ǣ� ��
A.��ˮ��![]() �����Һ©���Ǻò�����������ѹס��Һ©���ڲ���������ס�������֣��ѷ�Һ©����ת������
�����Һ©���Ǻò�����������ѹס��Һ©���ڲ���������ס�������֣��ѷ�Һ©����ת������
B.���ã�����Һ©����Һ��ֲ����ʹ��Һ©�����������ͨ�����ų�Һ�壩
C.��Һ©���Ļ�����ʹ�²�Һ��ȫ����ʢ��Һ����ձ��ڱ���������
D.��������������������ʢ�Ӳ������ϲ�Һ��
���𰸡�D
��������
A���ⲻ������ˮ�����������Ȼ�̼���ڷ�Һ©������ȡ����Һ����������ѹס��Һ©����������������ס�������֣��ѷ�Һ©����ת������Ȼ���ã�����������A��ȷ��
B��ʹ��Һ©�����������ͨ��ƽ��������ѹ����Һ�ֲ��֤Һ��˳��������B��ȷ��
C����Һ����ʱ����Һ©�����²�Һ����¿ڷų����²�Һ�����ʱ�����رջ������ϲ�Һ����Ͽڵ�������������Һ�����Ⱦ��C��ȷ��
D����Һ����ʱ����Һ©�����²�Һ����¿ڷų����²�Һ�����ʱ�����رջ������ϲ�Һ����Ͽڵ�������������Һ�����Ⱦ��D����
�ʴ�Ϊ��D��

����Ŀ�����ֶ�����Ԫ�������ڱ��е�λ����ͼ������ֻ��MΪ����Ԫ�ء�����˵��������ǣ� ��
Y | |||
M | X | Z |
A.����Z�����γ�Na2Z2������
B.��X������ɵ�����������ʱ�ܵ���
C.Y�������̬�⻯������ȶ��Ա�X��ǿ
D.M�ļ����Ӱ뾶��ͬ�����м����Ӱ뾶��С��
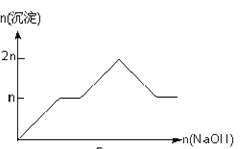
����Ŀ����MgCl2��AlCl3��Ϊnmol�Ļ����Һ�������е�NaOH��Һ������������NaOH�����ʵ��������ɳ��������ʵ����Ĺ�ϵ��ȷ���ǣ����ӣ������ʣ�����pH������
���� | Mg2+ | Al3+ | ���� | Al(OH)3 |
��ʼ����ʱ��pH | 8.93 | 3.56 | ��ʼ�ܽ�ʱ��pH | 8.04 |
��ȫ����ʱ��pH | 10.92 | 4.89 | ��ȫ�ܽ�ʱ��pH | 12.04 |
A. 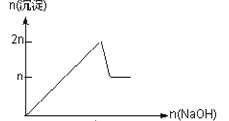
B. 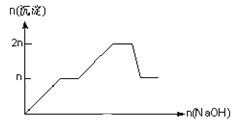
C. 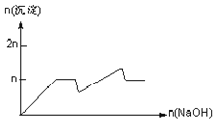
D.