��Ŀ����
����Ŀ��X��Y��Z��Q��WΪǰ��������ԭ���������������Ԫ�ء�Xԭ�ӵ�2p�ܼ���������δ�ɶԵ��ӣ����չ����Yԭ��K��ĵ�������M��ĵ������ij˻�������L��ĵ�������Z��Xͬ�壬Qλ�ڵڢ�B�壻WԪ��������ۺ�����۵Ĵ�����Ϊ4������������Ϣ���ش��������⡣
��1��WԪ�ص������ǣ�___________����ĵ����Ų�ʽΪ___________________��X�Ļ�̬ԭ�Ӻ�������ܼ�����_____�ֲ�ͬ�ռ��˶�״̬�ĵ��ӣ���֪������4QX��2Q2X+X2����Qԭ�Ӽ۲���ӽṹ�ĽǶȷ�����������Q2X��ԭ����__________________________________��
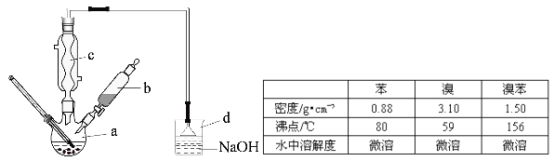
��2��Y��X�����γɶ��ָ��������ӣ�����ͼ��ʾ��������a��Ӧ�������ӻ�ѧʽΪYX![]() ����c��Ӧ�������ӵĻ�ѧʽΪ_________________(ԲȦ����Xԭ�ӣ��ڵ����Yԭ��)��
����c��Ӧ�������ӵĻ�ѧʽΪ_________________(ԲȦ����Xԭ�ӣ��ڵ����Yԭ��)��
��3����ʢ�к�Q2+����Һ�м��백ˮ�������������ܽ�����ӷ���ʽΪ___________________��д��������������ӵĽṹʽ____________________________________����λ����ע������
��4��W��Y��������Ԫ���γ���̬�⻯��������γɵ��������⻯���У�VSEPR����____________��������ͬ����������ͬ����������YH���й��õ��Ӷ�ƫ����Ԫ�أ�������W��Ӧʱ����W������������W��Y�ĵ縺����Դ�СΪ______________����Ԫ�ط������𣩡�
���𰸡��� [Ar] 3d104s24p4 3 CuO��ͭ����Χ�����Ų�Ϊ3d9��Cu2O��ͭ�ļ۵����Ų�Ϊ3d10�����ߴ���ȫ�������ȶ��ṹ����ǰ�߲��� Si6O![]() Cu(OH)2+4NH3��H2O= [Cu(NH3)4]2++2OH-+4H2O ����Cu(OH)2+4NH3= [Cu(NH3)4]2++2OH-��
Cu(OH)2+4NH3��H2O= [Cu(NH3)4]2++2OH-+4H2O ����Cu(OH)2+4NH3= [Cu(NH3)4]2++2OH-�� 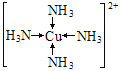 ��ͬ Se>Si
��ͬ Se>Si
��������
X��Y��Z��Q��WΪǰ��������ԭ���������������Ԫ�ء�Xԭ�ӵ�2p�ܼ���������δ�ɶԵ��ӣ����չ������X�ļ۵����Ų�ʽΪ2s22p4��XΪO��Yԭ��K��ĵ�������M��ĵ������ij˻�������L��ĵ�������K��2�����ӣ�L��8�����ӣ�����M���4�����ӣ�YΪSi��Qλ�ڵ���B����ֻ���ǵ�4���ڣ���QΪCu��Z��Xͬ�弴����A��X��Y��Z��Q��Wԭ����������������ZΪS��WԪ��������ۺ�����۵Ĵ�����Ϊ4��Wλ����A��ΪSe���ݴ˽�����֪ʶ�����ش�
(1)W��SeԪ�ص�����������ԭ������Ϊ34������ĵ����Ų�ʽΪ[Ar] 3d104s24p4����ԭ�ӹ�������ĸߵ��ɣ�X�Ļ�̬ԭ�Ӻ�������ܼ�Ϊ2p����3�ֲ�ͬ�ռ��˶�״̬�ĵ��ӣ���֪������4QX��2Q2X+X2��������������ͭ�ֽ�����������ͭ����������������Cu2Oԭ����CuO��ͭ����Χ�����Ų�Ϊ3d9��Cu2O��ͭ�ļ۵����Ų�Ϊ3d10�����ߴ���ȫ�������ȶ��ṹ����ǰ�߲��ǣ�
��Ϊ������[Ar] 3d104s24p4��3��CuO��ͭ����Χ�����Ų�Ϊ3d9��Cu2O��ͭ�ļ۵����Ų�Ϊ3d10�����ߴ���ȫ�������ȶ��ṹ����ǰ�߲��ǣ�
(2)Y��X�����γɶ��ָ��ӹ����������ӣ�������a��Ӧ�������ӻ�ѧʽΪYX![]() ��
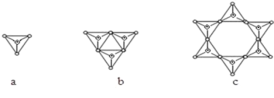
��![]() ���ǹ���������ṹ����c��Ӧ��������Ϊ6�����������幹�ɣ�����������ͨ�����ö�����ԭ�ӳ����������ģ���ÿ����������������1����ԭ�ӡ�2+2��
���ǹ���������ṹ����c��Ӧ��������Ϊ6�����������幹�ɣ�����������ͨ�����ö�����ԭ�ӳ����������ģ���ÿ����������������1����ԭ�ӡ�2+2��![]() =3����ԭ�ӣ� ��ѧʽΪSi6O
=3����ԭ�ӣ� ��ѧʽΪSi6O![]() ��
��
����Si6O![]() ��
��
(3)��ʢ�к�Q2+��ͭ���ӵ���Һ�м��백ˮ��������������Cu(OH)2������ܽ⣬������ܽ�����ӷ���ʽΪCu(OH)2+4NH3��H2O= [Cu(NH3)4]2++2OH-+4H2O����Cu(OH)2+4NH3= [Cu(NH3)4]2++2OH-������Ӧ��õ��������ӵĽṹʽΪ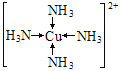 ��
��
����Cu(OH)2+4NH3��H2O= [Cu(NH3)4]2++2OH-+4H2O����Cu(OH)2+4NH3= [Cu(NH3)4]2++2OH-����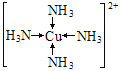 ��
��
(4)WΪSe��YΪSi����������Ԫ���γ���̬�⻯������γɵ�����⻯��ֱ�ΪH2Se��SiH
��Ϊ����ͬ��Se>Si��

 С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д� ��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�
��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�����Ŀ����֪��![]() ��������ͼװ�����������ϳ�����ȩ������������£�
��������ͼװ�����������ϳ�����ȩ������������£�

���� | �е�/�� | �ܶ�/��g�� | ˮ���ܽ��� |
������ | 117.2 | 0.8109 | �� |
����ȩ | 75.7 | 0.8017 | �� |
����˵���У���ȷ���� �� ��
A. ���õĴ�����ȩ�м������������ƣ����������Ƿ���������
B. ���¶ȼ�1ʾ��Ϊ90��95�棬�¶ȼ�2ʾ����117.2������ʱ���ռ�����
C. ��Ӧ������������ﵹ���Һ©�����Է�ȥˮ�㣬������ȩ�ӷ�Һ©���Ͽڵ���
D. Ϊ�ӿ췴Ӧ��Ӧ���ữ��Na2Cr2O7��Һһ����ȫ��������������