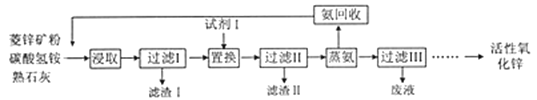
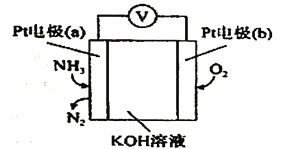
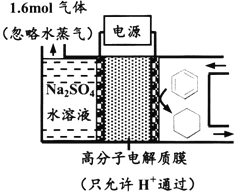
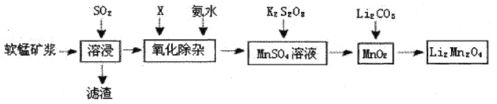
��Ŀ����
����Ŀ�����ܱ���Ϊ��߷��w���������Դ���������ʹ���������������õ���Ҫ�о�������ش��������⣺
��.������A(H3BNH3)��һ��DZ�ڵĴ�����ϣ�������Ԫ��״����(HB=NH)3ͨ���� �·�Ӧ�Ƶ�:3CH4+2 (HB=NH) 3+6H2O=3CO2+6H3BNH3
(1)��̬Bԭ�ӵļ۵����Ų�ʽΪ___________��B��C�� N�� O��һ�������ɴ�С��˳��Ϊ_________��CH4��H2O��CO2�ļ��ǰ����ɴ�С��˳������Ϊ___________��
(2)��(HB=NH)3����Ϊ�ȵ�������л�����Ϊ___________(�����ʽ)��
��.�����İ�ȫ���������������Ӧ�õĹؼ���
(1)ӡ�����³�Ƚ���ѧ������ĵ�Datta��Pati���˽���ADF������һ���� �ͻ�ϩ�ഢ�����(C16S8)������峣������۽Ƕ�֤�����ֲ��ϵķ��ӳ�ƽ��ṹ(��ͼ)��ÿ���ӻ�ƽ������������������10��H2���ӡ�
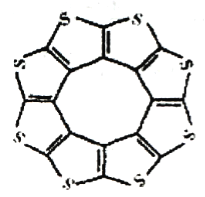
��C16S8������Cԭ�Ӻ�Sԭ�ӵ��ӻ�������ͷֱ�Ϊ___________��
����ؼ������������ʾ��
��ѧ�� | C-S | C=S | C16S8��̼��� |
����/pm | 181 | 155 | 176 |
�ӱ������ݿ�����C16S8��̼�����������C��S����C=S��֮�䣬ԭ�����______________________��
��C16S8��H2�������������___________��
(2)�д���ܵ�ͭ�Ͻ�������������ܶѻ��ṹ��������Cuԭ��λ�����ģ�Ag ԭ��λ�ڶ��㣬��ԭ�ӿɽ�����Cuԭ����Agԭ�ӹ��ɵ��������϶�С��þ��崢���ľ����ṹ��CaF2(��ͼ)���ƣ��þ��崢���Ļ�ѧʽΪ___________��

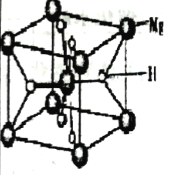
(3)MgH2�ǽ����⻯�ﴢ����ϣ��侧������ͼ��ʾ���þ�����ܶ�Ϊagcm-3���������Ϊ___________cm3(�ú�a��NA�Ĵ���ʽ��ʾ��NA��ʾ�����ӵ�������ֵ)��
���𰸡�2s22p1 N>O>C>B CO2>CH4>H2O sp2��sp3 C6H6 C16S8�����е�̼�������һ���̶ȵ�˫������ ���»��� Cu3AgH8 ![]()
��������
��(1)B��ԭ������Ϊ5�����ݵ����Ų�����1s22s22p1���۵����Ų�Ϊ2s22p1��B��C��N��Oͬ����Ԫ�أ����һ������Խ��Խ����Ҫ���ǵ�����״̬��N�ļ۵����Ų�Ϊ2s22p3��Ϊ���������ȶ��������ܻ�ϴ������Ų�ΪN>O>C>B�����ݷ��ӹ����Լ����ӶԻ������ۣ�CO2Ϊֱ���ͷ��ӣ�����Ϊ180�㣬CH4Ϊ�������壬����Ϊ109.5�㣬H2O��O�ŶԵ��ӻ�����O��H����ѹ�����ǻ�С��109.5�㣬���Լ��ǵ�����ΪCO2>CH4>H2O����Ϊ2s22p1 N>O>C>B CO2>CH4>H2O��
��2���ȵ����壬�۵��Ӻ�ԭ��������ȡ��л����к���C��ǡ��B��Nԭ�ӣ��ڼ۵��Ӻ�ԭ�����������2��Cԭ�ӣ���ȵ�����Ϊ(C2H2)3��ΪC6H6����ΪC6H6��
��(1) ��ÿ��Cԭ����Χ��3��Cԭ�ӣ���3���Ҽ���1���м���Cԭ�ӵ��ӻ�����Ϊsp2��ÿ��Sԭ���ܱ���2����������2�Թ¶Ե��ӣ��ӻ�����Ϊsp3����Ϊsp2��sp3��
��C16S8��̼�����������C��S����C=S��֮�䣬C16S8�����е�̼�������һ���̶ȵ�˫�����ʣ������ü������ڵ�����˫��֮�䣬��ΪC16S8�����е�̼�������һ���̶ȵ�˫�����ʣ�
��C16S8��H2��Ϊ���ӣ����Ӽ��������Ϊ���»�������Ϊ���»�����
(2) ������Cuԭ��λ�����ģ�Ag ԭ��λ�ڶ��㣬��ԭ�ӿɽ�����Cuԭ����Agԭ�ӹ��ɵ��������϶�У���ͼ�п��ĵ�ȦΪ���ص�Hԭ�ӣ����㾧���е�ԭ�ӣ�Cuԭ��λ�����ģ�Ϊ2�����������У���ÿ����������![]() ��Cuԭ�ӡ�ÿ��������Ag ԭ��λ�ڶ��㣬Ϊ8�����������У���
��Cuԭ�ӡ�ÿ��������Ag ԭ��λ�ڶ��㣬Ϊ8�����������У���![]() ��Agԭ�ӣ����ĵ�Ȧ�ھ����У�Ϊ��������ӵ�У�ÿ��������8��Hԭ�ӣ���ѧʽΪCu3AgH8����ΪCu3AgH8��
��Agԭ�ӣ����ĵ�Ȧ�ھ����У�Ϊ��������ӵ�У�ÿ��������8��Hԭ�ӣ���ѧʽΪCu3AgH8����ΪCu3AgH8��
(3)����![]() �����ݾ����ṹ��������������1��Mg��Ϊ�����������У� 8��������8��Mgԭ�ӣ�Ϊ8���������У���һ����
�����ݾ����ṹ��������������1��Mg��Ϊ�����������У� 8��������8��Mgԭ�ӣ�Ϊ8���������У���һ����![]() ��Mgԭ�ӡ������⻯�ﴢ����ϵĻ�ѧʽΪMgH2����ÿ��������2��Mgԭ�ӣ�4��Hԭ�ӡ�
��Mgԭ�ӡ������⻯�ﴢ����ϵĻ�ѧʽΪMgH2����ÿ��������2��Mgԭ�ӣ�4��Hԭ�ӡ�![]() ��
��![]() ��
��![]() ����Ϊ
����Ϊ![]() ��
��

����Ŀ���װ�(CH3NH2)�Ǻϳ�̫������������ԭ�ϡ�һ���¶��£������������Ϊ2.0 L�ĺ����ܱ������а���ͬ��ʽͶ�뷴Ӧ�������ӦCH3OH(g)��NH3(g) CH3NH2(g)��H2O(g)������й�ʵ���������£�
������� | �¶�/K | ��ʼ���ʵ�����mol�� | ƽ�����ʵ���mol | ||||
CH3OH | NH3 | CH3NH2 | H2O | CH3NH2 | H2O | ||
I | 530 | 0.40 | 0.40 | 0 | 0 | 0.30 | |
II | 530 | 0.80 | 0.80 | 0 | 0 | ||
III | 500 | 0 | 0 | 0.20 | 0.20 | 0.16 | |
����˵����ȷ����(����)
A. ����Ӧ��ƽ�ⳣ��K(��)��K(��)<K(��)
B. �ﵽƽ��ʱ����ϵ��c(CH3OH)��ϵ��2c(CH3OH����)>c(CH3OH����)
D. 530Kʱ������ʼ���������г���CH3OH 0.10 mol��NH3 0.15 mol��CH3NH2 0.10 mol��H2O 0.10 mol����Ӧ�����淴Ӧ�������