��Ŀ����
����ͼװ�ÿ��Խ��вⶨSO2ת����SO3��ת���ʵ�ʵ�顣��֪SO3���۵���16��8�棬�е���445��8�档��֪����װ�������漰��Ӧ�Ļ�ѧ����ʽΪ��
Na2SO3��s�� + H2SO4��98%��==Na2SO4 + H2O + SO2��

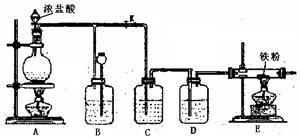
��1������ʵ����Ҫ��Ӧ���ڢ����Ӻ��ʵ�װ�á������ͼA��Eװ����ѡ�����ʺ�װ�ò����������������Ŀո��С�
�����ӵ�װ�÷ֱ���_______________��_______________��_______________��

��2�����Ҵ�����ͨ��O2��ΪʹSO2�нϸߵ�ת���ʣ�ʵ�����ڼ��ȴ�����μ�Ũ�����˳���У�Ӧ��ȡ�IJ����� ��
��3����SO2ͨ�뺬1��5mol�������Һ�У�������һ��ǿ���һ�����������1.5��6.02��1023������ת��ʱ���÷�Ӧ�Ļ�ѧ����ʽ
��4����amolNa2SO3��ĩ������Ũ������д�ʵ�飬����Ӧ����ʱ������ͨ��O2һ��ʱ����װ�â�������bg����ʵ����SO2��ת����Ϊ �����ú�a��b�Ĵ���ʽ��д��
��5��β�����θ���ܵ������� ��
Na2SO3��s�� + H2SO4��98%��==Na2SO4 + H2O + SO2��

��1������ʵ����Ҫ��Ӧ���ڢ����Ӻ��ʵ�װ�á������ͼA��Eװ����ѡ�����ʺ�װ�ò����������������Ŀո��С�
�����ӵ�װ�÷ֱ���_______________��_______________��_______________��

��2�����Ҵ�����ͨ��O2��ΪʹSO2�нϸߵ�ת���ʣ�ʵ�����ڼ��ȴ�����μ�Ũ�����˳���У�Ӧ��ȡ�IJ����� ��
��3����SO2ͨ�뺬1��5mol�������Һ�У�������һ��ǿ���һ�����������1.5��6.02��1023������ת��ʱ���÷�Ӧ�Ļ�ѧ����ʽ
��4����amolNa2SO3��ĩ������Ũ������д�ʵ�飬����Ӧ����ʱ������ͨ��O2һ��ʱ����װ�â�������bg����ʵ����SO2��ת����Ϊ �����ú�a��b�Ĵ���ʽ��д��
��5��β�����θ���ܵ������� ��
��1�� B A E��B A C��3�֣� ��2���ȼ��ȴ����ٵ���Ũ���� ��3�֣���3��SO2 + 2HClO3 = H2SO4 + 2ClO2��3�֣���4��  ��������
�������� �� 3�� ��5����ֹ�����е�CO2��ˮ�������ţ������� 2��
�� 3�� ��5����ֹ�����е�CO2��ˮ�������ţ������� 2��
 ��������
�������� �� 3�� ��5����ֹ�����е�CO2��ˮ�������ţ������� 2��
�� 3�� ��5����ֹ�����е�CO2��ˮ�������ţ������� 2����

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ��4��E����Ӧ��Ϻر�����K����ȥ�ƾ��ƣ��������ȵ����ã�A����������Cl2��������ʱB�е������� ��B�������� ��
��4��E����Ӧ��Ϻر�����K����ȥ�ƾ��ƣ��������ȵ����ã�A����������Cl2��������ʱB�е������� ��B�������� ��
 Fe2O3+SO3��SO3��+14H2O���������������ʵ��װ������֤�̷����ȷֽ��Ƿ���������Ӧ��
Fe2O3+SO3��SO3��+14H2O���������������ʵ��װ������֤�̷����ȷֽ��Ƿ���������Ӧ��
