��Ŀ����
16��ʵ��������500mL 0.2mol•L-1��Na2SO4��Һ��ʵ����������У�A�����Ƶõ���ҺС�ĵ�ת�Ƶ�����ƿ�У�
B������������ƿ�м�����ˮ��Һ���̶���1��2cm�������ý�ͷ�ι�С�ĵμ�����ˮ����Һ��Һ����ʹ���̶������У�
C������ƽ�ϳƳ�14.2g�����ƹ��壬���������ձ��У�������������ˮʹ����ȫ�ܽⲢ��ȴ�����£�
D������������ˮϴ���ձ��Ͳ�����2��3�Σ�ÿ��ϴ�ӵ�Һ�嶼С��ע������ƿ����������
E��������ƿƿ���������������µߵ���ҡ�ȣ�
����д���пհף�
��1�������������ȷ˳��Ϊ������ţ�CADBE��
��2����ʵ���õ��Ļ������������ձ�����ƽ�������롢���ӣ�������������ȱ�ٵ�������500ml����ƿ����ͷ�ιܡ�ҩ�ף�
��3�����������ʹ������ҺŨ��ƫ�ߵ��ǣ�����ţ�ad��
a������ʱ���Ӱ�Һ��
b��û���������IJ�������D
c��������ˮʱ�����������˿̶���
d��������մ������
e������ƿʹ��ǰ�ڱ�մ��ˮ�飮
���� ��1������������Һ��ʵ��������̽��в������������
��2������������Һ��ʵ���������ѡ������������
��3�������������������ʵ����ʵ�������Һ�������Ӱ�죬����C=$\frac{n}{V}$������������
��� �⣺��1������500ml0.2mol/L��Na2SO4��Һ�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ�����������ȷ��˳��Ϊ��CADBE��
�ʴ�Ϊ��CADBE��
��2������500ml0.2mol/L��Na2SO4��Һ�����������м��㡢�������ܽ⡢��Һ��ϴ����Һ�����ݡ�ҡ�ȵȲ������õ��������У�������ƽ��ҩ�ס����ӡ��ձ�����������500mL����ƿ����ͷ�ιܣ���ȱ�ٵ������У���������500ml����ƿ����ͷ�ιܣ�
�ʴ�Ϊ��500ml����ƿ����ͷ�ιܣ�ҩ�ף�
��3��a������ʱ���Ӱ�Һ�棬������Һ���ƫС����ҺŨ��ƫ�ߣ���ѡ��
b��û���������IJ�������D���������ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ��ʲ�ѡ��
c��������ˮʱ�����������˿̶��ߣ�������Һ���ƫ����ҺŨ��ƫ�ͣ��ʲ�ѡ��
d��������մ�����ʣ����³�ȡ�����ʵ�����ƫ�����ʵ����ʵ���ƫ����ҺŨ��ƫ�ߣ���ѡ��
e������ƿʹ��ǰ�ڱ�մ��ˮ�飬�����ʵ����ʵ�������Һ������������Ӱ�죬��ҺŨ�Ȳ��䣬�ʲ�ѡ��
��ѡ��ad��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���Ϥ���ƹ����ǽ���ؼ���ע����������������Ŀ�ѶȲ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�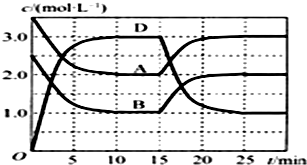 �¶�ΪTʱ����V L���ܱ������г���һ����������A��B��������Ӧ��A��g��+B��g��?C��s��+x D��g������H��0����Ӧ������������A��B��D�����ʵ���Ũ����ʱ��ı仯��ͼ��ʾ������˵����ȷ���ǣ�������
�¶�ΪTʱ����V L���ܱ������г���һ����������A��B��������Ӧ��A��g��+B��g��?C��s��+x D��g������H��0����Ӧ������������A��B��D�����ʵ���Ũ����ʱ��ı仯��ͼ��ʾ������˵����ȷ���ǣ�������| A�� | �÷�Ӧ��ƽ�ⳣ������ʽK=$\frac{c��C��•{c}^{2}��D��}{c��A��•c��B��}$ | |
| B�� | ��ƽ��ʱ�����¶Ȳ��䣬ѹ���������ƽ�����淴Ӧ�����ƶ� | |
| C�� | ��Ӧ��15 minʱ���ı�������������¶� | |
| D�� | ��Ӧ��ǰ10 min��ƽ����Ӧ���ʣ�v��D��=0.3 mol•L-1•min-1 |
 ����EVA
����EVA��������ϩһ������ϩ��������ʣ�����������ֳ���ԣ�180�濪ʼ���ַֽ�����CO��NH3���˺��۾���Ƥ���Ĥ������˵����ȷ���ǣ�
| A�� | EVA�������ڸ߷��ӻ�������� | |
| B�� | NH3����ˮ�ܵ��磬����NH3�ǵ���� | |
| C�� | ��������Ħ������Ϊ45 | |
| D�� | �������ֽ�����CO��NH3����������ԭ��Ӧ |
 ��ͼ��ƿ��װ����c mol•L��1��Na2SO4��Һ�����ⶨ���к���a mol Na+������ƿ��
��ͼ��ƿ��װ����c mol•L��1��Na2SO4��Һ�����ⶨ���к���a mol Na+������ƿ����Ϊ��������
| A�� | $\frac{a}{c}$ L | B�� | $\frac{a}{2c}$L | C�� | ac L | D�� | 2ac L |
| A�� | �ٺ͢�һ���� | B�� | �ڱȢٶ� | C�� | �ٱȢڶ� | D�� | ���Ƚ� |