��Ŀ����
�±���Ԫ�����ڱ���һ����, ��Ա��еĢ١�����Ԫ��,��д���пհ�:
(1)����ЩԪ����,��ѧ��������õ���: (�����Ԫ�ط��Ż�ѧʽ,��ͬ)��
(2)��Ԫ������������ˮ�����У�������ǿ����____ ___��������ǿ����______ _��
(3)д������ݵ�����������Ӧˮ���ﻥ�෴Ӧ�����ӷ���ʽ__________ _ ��
(4)�õ���ʽ��ʾԪ�آ�����γɵĻ����
(5)��ʾ��������̬�γɵĻ�����ṹʽ ��
(6)д��Ԫ�آڵ�����������ˮ�����������̬�⻯�ﷴӦ�Ļ�ѧ����ʽ
(7)�ۡ�����Ԫ���γɵ��⻯��е�ϸߵ��� ���û�ѧʽ��ʾ������ԭ���� ��
| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0�� |
| 2 | | | | �� | �� | �� | | |
| 3 | �� | | �� | | | �� | �� | �� |
| 4 | �� | | | | | | | |
(2)��Ԫ������������ˮ�����У�������ǿ����____ ___��������ǿ����______ _��
(3)д������ݵ�����������Ӧˮ���ﻥ�෴Ӧ�����ӷ���ʽ__________ _ ��
(4)�õ���ʽ��ʾԪ�آ�����γɵĻ����
(5)��ʾ��������̬�γɵĻ�����ṹʽ ��
(6)д��Ԫ�آڵ�����������ˮ�����������̬�⻯�ﷴӦ�Ļ�ѧ����ʽ
(7)�ۡ�����Ԫ���γɵ��⻯��е�ϸߵ��� ���û�ѧʽ��ʾ������ԭ���� ��
����14�֣���1��Ar ��2�֣� (2)HClO4 KOH ��2�֣�
(3)OH-+Al(OH)3��AlO2-+2H2O ��2�֣� (4) ��2�֣�
��2�֣�
��5��O��C��O ��2�֣� ��6��NH3+HNO3��NH4NO3 ��2�֣���7��H2O , ˮ���Ӽ������� ��2�֣�
(3)OH-+Al(OH)3��AlO2-+2H2O ��2�֣� (4)
 ��2�֣�
��2�֣���5��O��C��O ��2�֣� ��6��NH3+HNO3��NH4NO3 ��2�֣���7��H2O , ˮ���Ӽ������� ��2�֣�
�������������Ԫ���������ڱ��е�λ�ÿ�֪���١�����Ԫ�طֱ���C��N��O��Na��Al��S��Cl��Ar��K��
(1)����Ԫ�������ɿ�֪������Ԫ��,��ѧ��������õ���Ar��
(2)�ǽ����Ի������Խǿ������������ˮ��������Ի����Խǿ����������������ˮ���������ǿ����KOH��������ǿ����HClO4��
(3)����������������������������Ԣ���ݵ�����������Ӧˮ���ﻥ�෴Ӧ�����ӷ���ʽ��OH-+Al(OH)3��AlO2-+2H2O��
(4)Ԫ�آ�����γɵĻ����������ƣ��������Ӽ������ӻ��������ʽ��
 ��
��(5)��������̬�γɵĻ�������CO2�����й��ۼ��Ĺ��ۻ�����ṹʽ��O��C��O��
(6)Ԫ�آڵ�����������ˮ�����������̬�⻯�ﷴӦ�Ļ�ѧ����ʽNH3+HNO3��NH4NO3��
(7)ˮ���Ӽ����������е�ߣ����Ԣۡ�����Ԫ���γɵ��⻯��е�ϸߵ���H2O��
�����������Ǹ߿��еij������㣬���ڻ���������Ŀ��顣���ض�ѧ������֪ʶ�Ĺ�����ѵ��������������ѧ����������������������Ҫ��Ԫ�ء�λ�������ԡ����߹�ϵ���ۺϿ��飬�Ƚ�ȫ�濼��ѧ���й�Ԫ���ƶ�֪ʶ���������֪ʶ�������������ԡ����ڱ���Ԫ�ص��ƶϡ�Ϊ���壬����ѧ����Ԫ�����ڱ�����Ϥ�̶ȼ���Ա��и�Ԫ�����ʺ���Ӧԭ�ӽṹ�������Եݱ���ɵ���ʶ�����ճ̶ȡ�

��ϰ��ϵ�д�
 ��ѧ�̸̳����¿α�ϵ�д�
��ѧ�̸̳����¿α�ϵ�д� Сѧ��ʱ��ѵϵ�д�
Сѧ��ʱ��ѵϵ�д�
�����Ŀ


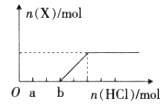
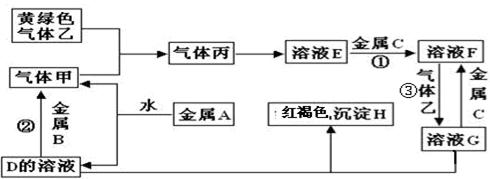
 ��2.5��
��2.5�� ���ֽ��ó�������0.5 mol��
���ֽ��ó�������0.5 mol��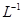 ��BaCl2��Һ�У���
��BaCl2��Һ�У���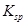 ________�����������С�����䡱������ɲ�����F������������Һ�е�Ũ��Ϊ________mol��
________�����������С�����䡱������ɲ�����F������������Һ�е�Ũ��Ϊ________mol�� ��
�� �����������
�����������