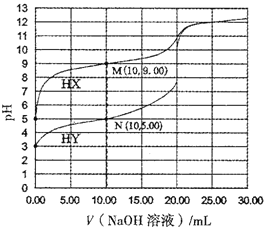
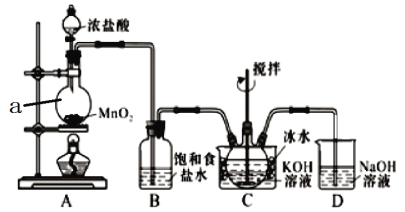
��Ŀ����
����Ŀ��������ͼװ�òⶨ�к��ȵ�ʵ�鲽�����£�

������Ͳ��ȡ50mL 0.25mol/L���ᵹ��С�ձ��У���������¶ȣ�
������һ��Ͳ��ȡ50mL 0.55mol/L NaOH��Һ��������һ�¶ȼƲ�����¶ȣ�
����NaOH��Һ����С�ձ��У��跨ʹ֮��Ͼ��ȣ�������Һ������¶ȡ�
�ش��������⣺
��1������A������Ϊ_________________________��
��2������NaOH��Һ����ȷ������________��
A���ز����������� B���������������� C��һ��Ѹ�ٵ���
��3��ʹ������NaOH��Һ��Ͼ��ȵ���ȷ������________��
A�����¶ȼ�С�Ľ��� B���ҿ�ӲֽƬ�ò���������
C����������ձ� D���������¶ȼ��ϵĻ��β���������ؽ���
��4��ʵ���������±���
������д�±��еĿհף�
�¶� ʵ�� ���� | ��ʼ�¶�t1/�� | ��ֹ �¶� t2/�� | �¶Ȳ� ƽ��ֵ (t2��t1)/�� | ||
H2SO4 | NaOH | ƽ��ֵ | |||
1 | 26.2 | 26.0 | 26.1 | 29.5 | _______ |
2 | 27.0 | 27.4 | 27.2 | 32.3 | |
3 | 25.9 | 25.9 | 25.9 | 29.2 | |
4 | 26.4 | 26.2 | 26.3 | 29.8 | |
�����Ƶ���Ϊ0.55mol/L NaOH��Һ��0.25mol/L������Һ���ܶȶ���1 g/cm3���кͺ�������Һ�ı�����c��4.18 J/(g����)�����к�����H �� ______ (ȡС�����һλ)��
���к��Ȳⶨʵ���У����в���һ���ή��ʵ��ȷ�Ե���________��
A���õζ���(��������������������0.01)ȡ���������Һ�����
B��NaOH��Һ�ڵ���С�ձ�ʱ������������
C����С�ձ�������ϴв��ŵ�����ĭ���Ͻ϶�
D������HCl��Һ���¶ȼ���ˮϴ�����������NaOH��Һ���¶�
���𰸡����β�������� C D 3.4 ��56.8kJ/mol B
��������
��1������װ��ͼ��������A��������(2)��NaOH��Һ����С�ձ��У��ּ��ε��룬�ᵼ������ɢʧ��Ӱ��ⶨ�����(3)������������ƻ��ʱ���������¶ȼ��ϵĻ��β������������ؽ�����ʹ������NaOH��Һ��Ͼ����� (4) �ٵ�һ��ʵ����¶Ȳ���3.4���ڶ���ʵ����¶Ȳ���5.1��������ʵ����¶Ȳ���3.3�����Ĵ�ʵ����¶Ȳ���3.5�����ڵڶ���ʵ������ƫ������ֵ��ֻ�ܸ��ݵ�1��3��4��ʵ������¶Ȳ��ƽ��ֵ���ڸ��ݼ��㹫ʽQ=cm��T����ʵ��ų����������к�����ǿ��ǿ��ϡ��Һ��ȫ��Ӧ����1molˮ�ų�����������NaOH��Һ�ڵ���С�ձ�ʱ��������������ʵ��������ˮ�������٣��ų���������С������к��ȵ���ֵ����
��1������װ��ͼ������A�������ǻ��β����������(2)��������������Һʱ������һ��Ѹ�ٵĵ��룬Ŀ���Ǽ���������ɢʧ�����ּܷ��ε�������������Һ������ᵼ������ɢʧ��Ӱ��ⶨ�������ѡC�� (3)ʹ������NaOH��Һ��Ͼ��ȵ���ȷ����������:�������¶ȼ��ϵĻ��β������������ؽ������¶ȼ��Dz����¶ȵģ�����ʹ���¶ȼƽ�����Ҳ������������ձ���������ܵ���Һ�彦��������ɢʧ��Ӱ��ⶨ����������ܴ�ӲֽƬ�ò��������裬�����������ɢʧ����ѡD��(4) �ٵ�һ��ʵ����¶Ȳ���3.4���ڶ���ʵ����¶Ȳ���5.1��������ʵ����¶Ȳ���3.3�����Ĵ�ʵ����¶Ȳ���3.5�����ڵڶ���ʵ������ƫ������ֵ��ֻ�ܸ��ݵ�1��3��4��ʵ������¶Ȳ��ƽ��ֵ��![]() ����50mL��0.50mol/L������50mL��0.55mol/L����������Һ��������Ϊm=100mL��1g/cm3=100g��c=4.18 J/(g����)�����빫ʽQ=cm��T������0.025mol��ˮ�ų�����Q=4.18 J/(g����)��100g��3.4��=1.42kJ��������0.025mol��ˮ�ų�����Ϊ1.42kJ����������1mol��ˮ�ų�����Ϊ1.42kJ��
����50mL��0.50mol/L������50mL��0.55mol/L����������Һ��������Ϊm=100mL��1g/cm3=100g��c=4.18 J/(g����)�����빫ʽQ=cm��T������0.025mol��ˮ�ų�����Q=4.18 J/(g����)��100g��3.4��=1.42kJ��������0.025mol��ˮ�ų�����Ϊ1.42kJ����������1mol��ˮ�ų�����Ϊ1.42kJ��![]() =56.8kJ������ʵ���õ��к��ȡ�H=-56.8kJ/mol����A���õζ��ܣ���������������������0.01��ȡ���������Һ��������������ȷ�����ʵ��ȷ�ԣ� B������������Һ����ʱ����������٣���ʹ���к�������ƫС������һ���ή��ʵ��ȷ�ԣ� C����С�ձ��в��ŵ�����ĭ���Ͻ϶࣬����Ч�����ã������ʵ��ȷ�ԣ� D������HCl��Һ���¶ȼ���ˮϴ���ٲ��������ƣ��������ͼ�֮����Ϊ�кͷ�Ӧ�����µ�������ʧ�����ʵ���ȷ��������������B�������⣬ѡB.
=56.8kJ������ʵ���õ��к��ȡ�H=-56.8kJ/mol����A���õζ��ܣ���������������������0.01��ȡ���������Һ��������������ȷ�����ʵ��ȷ�ԣ� B������������Һ����ʱ����������٣���ʹ���к�������ƫС������һ���ή��ʵ��ȷ�ԣ� C����С�ձ��в��ŵ�����ĭ���Ͻ϶࣬����Ч�����ã������ʵ��ȷ�ԣ� D������HCl��Һ���¶ȼ���ˮϴ���ٲ��������ƣ��������ͼ�֮����Ϊ�кͷ�Ӧ�����µ�������ʧ�����ʵ���ȷ��������������B�������⣬ѡB.

����Ŀ���״���һ�ֿ�������Դ����CO2�Ʊ��״��Ĺ��̿����漰�ķ�Ӧ���£�
��Ӧ����CO2(g)+3H2(g)![]() CH3OH(g)+H2O(g) ��H1=��49.58 kJmol��1
CH3OH(g)+H2O(g) ��H1=��49.58 kJmol��1
��Ӧ����CO2(g)+H2(g)![]() CO(g)+H2O(g) ��H2
CO(g)+H2O(g) ��H2
��Ӧ����CO(g)+2H2(g)![]() CH3OH(g) ��H3=��90.77 kJmol��1
CH3OH(g) ��H3=��90.77 kJmol��1
�ش��������⣺
(1)��Ӧ��ġ�H2=_________������Ӧ����������ƽ�ⳣ���ֱ�ΪK1��K2��K3����K2=________(��K1��K3��ʾ)��
(2)��Ӧ���Է�����������___________(�����ϵ��¶��������ϸ��¶��������κ��¶���)��
(3)��һ��������2 L�����ܱ������г���3 mol H2��1.5 mol CO2����������Ӧ��ʵ���ò�ͬ��Ӧ�¶�����ϵ��CO2��ƽ��ת���ʵĹ�ϵ�����±���ʾ��
�¶�(��) | 500 | T |
CO2��ƽ��ת���� | 60�G | 40�G |
��T______500��(����>������<�� ����=��)��
���¶�Ϊ500��ʱ���÷�Ӧ10 minʱ�ﵽƽ�⡣��H2��ʾ�÷�Ӧ�ķ�Ӧ����v(H2)=______________�����¶��£���ӦI��ƽ�ⳣ��K=______________L2/mol2
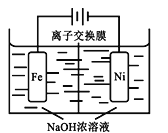
(4)��CO2�Ʊ��״�����Ҫ��������ҵ���õ�ⷨ��ȡNa2FeO4��ͬʱ���������Fe+2H2O+2OH![]() FeO42+3H2��������ԭ����ͼ��ʾ�����һ��ʱ���c(OH)���͵�������__________ (������������������������)�����ҷ����ĵ缫��ӦʽΪ��___________________________��
FeO42+3H2��������ԭ����ͼ��ʾ�����һ��ʱ���c(OH)���͵�������__________ (������������������������)�����ҷ����ĵ缫��ӦʽΪ��___________________________��