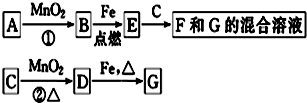
��Ŀ����
17��ij��ҵ��ˮ�н����±������е�5�֣�������ˮ�ĵ��뼰���ӵ�ˮ�⣩���Ҹ������ӵ����ʵ���Ũ����ȣ���Ϊ0.1mol/L��| ������ | K+ Cu2+ Fe3+ Al3+ Fe2+ |
| ������ | Cl- CO32-NO3-SO42-SiO32- |
���ò�˿պȡ������Һ���ڻ��������գ�����ɫ���棨����ɫ�ܲ����۲죩��
��ȡ������Һ������KSCN��Һ�����Ա仯��
����ȡ��Һ�����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ����ʱ��Һ��Ȼ���壬����Һ��������������ԭ��Һ��������ͬ��
������������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɣ�
���ƶϣ�
��1�����ɢ��жϣ���Һ��һ�������е���������K+��Fe3+��д���ӷ��ţ���
��2�����м�����������������ɫ��������ӷ���ʽ��3Fe2++NO3-+4H+�T3Fe3++NO��+2H2O��
��3�����������ú���ɫ����ͨ��ˮ�У��������ɫ���������Ļ�ѧ����ʽΪ3NO2+H2O�T2HNO3+NO��
��4����ͬѧ����ȷ��ԭ��Һ��������������Fe2+��Cu2+����������Cl-��NO3-��SO42-����д���ӷ��ţ�
��5����ҵ��ˮ�г����в�ͬ���͵���Ⱦ��ɲ��ò�ͬ�ķ�����������������ͬѧ��Ժ���ͬ��Ⱦ��ķ�ˮ����Ĵ�����ʩ�ͷ�����������ȷ����D��
| ѡ�� | ��Ⱦ�� | ������ʩ | ������� |
| A | ���� | ����ʯ���к� | ������ |
| B | Cu2+���ؽ������� | �������γ��� | ��ѧ�� |
| C | �������л���ķ�ˮ | ͨ�������л | ������ |
| D | ���Եķ�ˮ | ��CO2���к� | ��ѧ�� |
���� ���ò�˿պȡ������Һ���ڻ��������գ�����ɫ���棨����ɫ�ܲ����۲죩��˵��û��K+��
��ȡ������Һ������KSCN��Һ�����Ա仯��˵��û��Fe3+��
����ȡ��Һ�����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ����ʱ��Һ��Ȼ���壬����Һ������������䣬˵�� Fe2+��NO3-��H+��Ӧ����NO������Һ����Fe2+��NO3-����������Һ������������䣬˵��ԭ��Һ����Cl-����������Һ��Ȼ���������˵��û��SiO32-��
������������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɣ�˵����SO42-���Դ������1������4����
��5����Ⱦ����Ҫ���ܳ����ж������ʣ������ɵ����ʶԻ�������Ⱦ���������뻯ѧ���������Ƿ������������ɣ�
��� �⣺���ò�˿պȡ������Һ���ڻ��������գ�����ɫ���棨����ɫ�ܲ����۲죩��˵��û��K+��
��ȡ������Һ������KSCN��Һ�����Ա仯��˵��û��Fe3+��
����ȡ��Һ�����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ����ʱ��Һ��Ȼ���壬����Һ������������䣬˵�� Fe2+��NO3-��H+��Ӧ����NO������Һ����Fe2+��NO3-����������Һ������������䣬˵��ԭ��Һ����Cl-����������Һ��Ȼ���������˵��û��SiO32-��
������������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɣ�˵����SO42-��
��1���ɢ��жϣ���Һ��һ�������е���������K+��Fe3+���ʴ�Ϊ��K+��Fe3+��
��2�����м�����������������ɫ���壬��Fe2+��NO3-��H+��Ӧ����NO�������ӷ���ʽ��3Fe2++NO3-+4H+�T3Fe3++NO��+2H2O��
�ʴ�Ϊ��3Fe2++NO3-+4H+�T3Fe3++NO��+2H2O��
��3�����������ú���ɫ����Ϊ����������ͨ��ˮ�У���ˮ��Ӧ����NO���������ɫ���������Ļ�ѧ����ʽΪ3NO2+H2O�T2HNO3+NO��
�ʴ�Ϊ��3NO2+H2O�T2HNO3+NO��
��4���������ƶϿ�֪��Һ��������Ϊ Cl-��NO3-��SO42-���Ҹ�Ϊ0.1mol/L���Ѿ��ƶϳ�����������Fe2+����Ũ��Ϊ0.1mol/L���ɵ���غ��֪��Һ�л���һ��+2�������ӣ����Ի���Cu2+�����Լ�ͬѧ����ȷ��ԭ��Һ�������������ǣ�Fe2+��Cu2+���������ǣ�Cl-��NO3-��SO42-��
�ʴ�Ϊ��Fe2+��Cu2+��Cl-��NO3-��SO42-��
��5��A����ͼ���кͷ�ӦΪ��ѧ��Ӧ��Ϊ��ѧ������A����
B������ͭ���ܣ����Բ��ܳ�����ȥ����B����
C�������л���ͨ�������л�Ĺ����ǻ�ѧ��������C����
D��������̼���������壬���Ժͼ�֮�䷴Ӧ�����ڻ�ѧ��������D��ȷ��
��ѡD��
���� ���⿼�����Ӽ�������ƶϣ�Ϊ��Ƶ���㣬��������֮��ķ�Ӧ�����Ӽ��鼰��������۵Ĺ�ϵΪ���Ĺؼ������ط������ƶ������Ŀ��飬��Ŀ�ѶȲ���

| A�� | ������Һ�ʹ���ʯ�е�CaCO3��Ӧ��CaCO3+2H+�TCa2++CO2��+H2O | |
| B�� | ��������Һ��ͨ��������CO2��C6H5O-+CO2+H2O-��C6H5OH+HCO3- | |
| C�� | ����ʯ��ˮ�����NaHCO3��Һ��Ӧ��HCO3-+Ca2++OH-�TCaCO3��+H2O | |
| D�� | ����ˮ��Һ�ʼ��Ե�ԭ��S2-+2H2O?H2S��+2OH- |
| A�� | ���¸ֹ�����þ�� | B�� | ������������Ϳ�� | ||
| C�� | ��ˮ������Ϳ�߷���Ĥ | D�� | ˮ�еĸ�բ�����ӵ�Դ�ĸ��� |
| A�� | 5 s | B�� | ����5 s | C�� | С��5 s | D�� | ���ж� |
| A�� | Cl2 | B�� | O3 | C�� | NH3 | D�� | KOH |
| ���ѻ����ɵĻ�ѧ�� | �������� |
| ����1mol H2�����еĻ�ѧ�� | ��������436kJ |
| ����1mol I2�����еĻ�ѧ�� | ��������151kJ |
| �γ�1mol HI�����еĻ�ѧ�� | �ͷ�����299kJ |
| A�� | �÷�ӦΪ���ȷ�Ӧ | |
| B�� | �÷�Ӧ�з�Ӧ������������������������ | |
| C�� | HI�����еĻ�ѧ����H2�����еĻ�ѧ�����ι� | |
| D�� | ���ж� |
| A�� | ����ķ���ʽCH3COOH | |
| B�� | ��ϩ�Ľṹ��ʽCH2CH2 | |
| C�� | ��������ģ��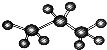 | |
| D�� | �Ҵ��Ĺ�������-OH����һ����λ����� |